Ammonium hexachlorostannate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
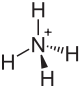 
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ammonium hexachlorostannate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | (NH 4 ) 2 [SnCl 6 ] | |||||||||||||||
| Brief description |
white solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 367.49 g · mol -1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.387 g cm −3 |
|||||||||||||||
| solubility |
soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Ammonium hexachlorostannate is the ammonium salt of hexachlorotin (IV) acid H 2 [SnCl 6 ].
Extraction and presentation
Ammonium hexachlorostannate is formed from tin (IV) chloride (stannous chloride ) and ammonium chloride (salmiac).
properties
Ammonium hexachlorostannate is a white solid that is soluble in water.
The compound crystallizes cubic , space group Fm 3 m (space group no. 225) , with the lattice parameter a = 10.04 Å .
use
Due to its use for dyeing (as a stain ), it is also referred to by the technical name of pink salt ( pink = pink). The neutral reacting salt is used where the tin (IV) chloride solution containing free acid cannot be used.
It accelerates the development of the tin plague .
Individual evidence
- ↑ a b c d e data sheet Ammonium hexachlorostannate (IV), Puratronic®, 99.999% (metals basis) from AlfaAesar, accessed on December 7, 2019 ( PDF )(JavaScript required) .
- ↑ a b Heinrich Remy: Textbook of Inorganic Chemistry Volume I, Leipzig 1970, page 650
- ^ A b Lueger: Lexicon of the entire technology . 2nd Edition. 1904, entry [1003] tin chlorides. ( Web digitization , zeno.org).
- ↑ G. Engel: The crystal structures of some hexachloro complex salts . In: Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie, 90, 1935, pp. 341–373 doi: 10.1524 / zkri.1935.90.1.341 .
- ↑ Pink Salt . In: Heinrich August Pierer , Julius Löbe (Hrsg.): Universal Lexicon of the Present and the Past . 4th edition. tape 13 . Altenburg 1861, p. 144 ( zeno.org ).
- ↑ tin chloride . In: Meyers Konversations-Lexikon . 4th edition. Volume 16, Verlag des Bibliographisches Institut, Leipzig / Vienna 1885–1892, p. 924.

![{\ displaystyle \ mathrm {SnCl_ {4} +2 \ NH_ {4} Cl \ longrightarrow (NH_ {4}) _ {2} [SnCl_ {6}]}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3ac4a0a93dae5cebdb4f2387252e5c4e5de13ef3)