Ammonium hydrogen sulfite
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
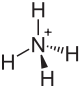 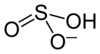
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ammonium hydrogen sulfite | |||||||||||||||
| other names |
Ammonium bisulfite |
|||||||||||||||
| Molecular formula | (NH 4 ) HSO 3 | |||||||||||||||
| Brief description |
colorless to yellowish solid with a smell of sulfur dioxide |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 99.11 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.03 g cm −3 |
|||||||||||||||
| Melting point |
50 ° C (decomposition) |
|||||||||||||||
| Sublimation point |
150 ° C (in nitrogen) |
|||||||||||||||
| solubility |
easily in water (2670 g l −1 at 10 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Ammonium hydrogen sulfite is an inorganic chemical compound from the group of ammonium salts and sulfites .
Extraction and presentation
Ammonium hydrogen sulfite is produced, for example, in flue gas desulfurization in the Walter process by injecting ammonia into the exhaust gas flow.
properties
Ammonium hydrogen sulfite is a colorless to yellowish, deliquescent solid with a smell of sulfur dioxide . It is only traded as an aqueous solution. The compound decomposes when exposed to acids with formation of sulfur dioxide, when exposed to alkalis ammonia .
use
Ammonium hydrogen sulfite is used for the sulfurization of wine and also serves as a preservative.
Individual evidence
- ↑ a b c d e f g h i Entry on ammonium hydrogen sulphite in the GESTIS substance database of the IFA , accessed on January 26, 2015(JavaScript required) .
- ↑ Michael Schultes: Exhaust gas cleaning: process principles, calculation bases, process comparison . Springer-Verlag, 2011, ISBN 978-3-642-18191-7 , pp. 74 ( limited preview in Google Book search).
- ^ Klaus Rädle: Champagne: facts, data, background . book-on-demand.de, 2009, ISBN 978-3-86805-327-2 , p. 74 ( limited preview in Google Book search).
- ↑ Marina Bährle-Rapp: Springer Lexicon Cosmetics and Body Care . Springer-Verlag, 2013, ISBN 978-3-662-09983-4 ( limited preview in Google book search).

