Aucubin
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
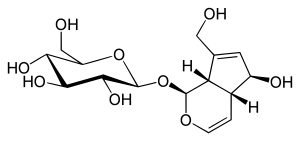
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Aucubin | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 15 H 22 O 9 | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 346.33 g · mol -1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
180–184 ° C (decomposition) |
|||||||||||||||
| solubility |
partially soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Aucubin is a common phytochemical from the group of iridoids . It is a glycoside of the aglycone Aucubigenin with a unit of glucose . Like other monoterpenes , the aglycone is synthesized from two isoprene units with five carbon atoms each in the plant. In contrast to the C10 basic body of the iridoids, the aucubin skeleton has only nine carbon atoms, as a methyl group is oxidized to a carboxylic acid group during biosynthesis and then split off as carbon dioxide .
Occurrence and use
Aucubin has an antibiotic effect and affects blood pressure. The juice of plantain contains Aucubin and not moldy. Most other plants' juices go moldy. Aucubin has anti-inflammatory and soothing effects. The itching and swelling of insect bites can be reduced by applying crushed ribwort leaves. The semi-parasitic eyebright and the mullein (woolly flower) also contain aucubin - like the speedwell ( Veronica officinalis ). They have an anti-inflammatory effect. However, the aucubin is only retained with very careful extraction. The aucubin has often lost ribwort due to improper drying or storage. Aucubin makes the tea bitter. Since it is broken down by intestinal bacteria, it has no pharmacological effect in tea.
literature
- R. Hänsel, O. Sticher: Pharmakognosie, Phytopharmazie , 8th edition, Springer Verlag, Berlin 2007, ISBN 3-540-34256-7 .
- J. Falbe, M. Regitz: Römp Chemie Lexikon Vol. 1 , 9th edition, Georg Thieme Verlag 1995, ISBN 3-13-102759-2
- Ruprecht Düll , Herfried Kutzelnigg : Pocket dictionary of plants in Germany. A botanical-ecological excursion companion to the most important species. 6th, completely revised edition. Quelle & Meyer, Wiebelsheim 2005, ISBN 3-494-01397-7 .
- C. Jänicke, J. Grünwald, T. Brendler: Handbuch Phytotherapie , Wissenschaftliche Verlagsgesellschaft mbH, Stuttgart 2003, ISBN 3-8047-1950-3
Individual evidence
- ↑ a b c d data sheet Aucubin at Sigma-Aldrich , accessed on February 7, 2019 ( PDF ).
- ↑ Indofine Chemical Company: Aucubin with HPLC | 479-98-1 | INDOFINE Chemical Company , accessed June 28, 2019
- ↑ Qing-Qing Wu, Yang Xiao et al. a .: Aucubin protects against pressure overload-induced cardiac remodeling the β-adrenoceptor-neuronal NOS cascades. In: British Journal of Pharmacology. 175, 2018, p. 1548, doi : 10.1111 / bph.14164 .
