Catechol violet
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
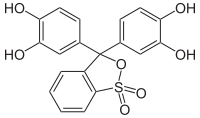
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Catechol violet | |||||||||||||||
| Molecular formula | C 19 H 14 O 7 S | |||||||||||||||
| Brief description |
dark green odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 386.36 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
185 ° C |
|||||||||||||||
| solubility |
bad in water (1 g l −1 ) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Pyrocatechol violet is a chemical compound from the group of triphenylmethane dyes . It is used as an indicator substance which changes its color from red in the pH range <0, via yellow in the range from 2–8 to purple in the range around 8 and red-violet in the basic range. It forms complex compounds with metal ions .
Extraction and presentation
Catechol violet can be obtained by reacting catechol with 2-sulfobenzoic anhydride .
use
Catechol violet solutions (e.g. EDTA ) can be used for the determination of many metals ( titration for chelates and other complexes ), especially for the detection of tin . The chelates formed by metals with catechol violet are generally blue to blue-violet in color.
Individual evidence
- ↑ a b c data sheet Catechol Violet at AlfaAesar, accessed on December 7, 2019 ( PDF )(JavaScript required) .
- ↑ a b c Datasheet Pyrocatechol Violet, suitable for indicator at Sigma-Aldrich , accessed on December 7, 2019 ( PDF ).
- ↑ Google Patents: CN103387563A - Preparation method for catechol violet - Google Patents , accessed December 7, 2019