Cesium tungstate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
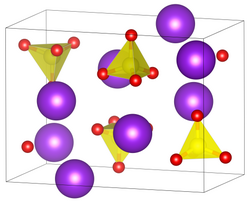
| __ Cs + __ W 6+ __ O 2− | ||||||||||||||||
| Crystal system |
orthorhombic |
|||||||||||||||
| Space group |
Pmcn (No. 62, position 5) |
|||||||||||||||
| Lattice parameters |
|
|||||||||||||||
| General | ||||||||||||||||
| Surname | Cesium tungstate | |||||||||||||||
| other names |
Cesium tungsten oxide |
|||||||||||||||
| Molecular formula | Cs 2 WO 4 | |||||||||||||||
| Brief description |
white odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 513.66 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| solubility |
soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Cesium tungstate is an inorganic chemical compound of cesium from the group of tungstates .
Extraction and presentation
Examples of possible reactions are:
- made of tungsten (VI) oxide and cesium hydroxide (analogous to the representation of sodium tungstate ):
- from cesium chloride and silver tungstate :
properties
Cesium tungstate is a white, odorless solid that is soluble in water. At room temperature the compound is present in an orthorhombic crystal system in the space group Pmcn (space group no. 62, position 5) isotypic to cesium molybdate and β- potassium sulfate . At 536 ° C there is a phase change from an orthorhombic to a hexagonal crystal structure .
use
Cesium tungstate is used to produce transparent insulation coatings with good properties for infrared radiation .
Individual evidence
- ↑ a b F. Kools X nm, AS Koster, GD Rieck: The Structures of potassium, rubidium and cesium molybdates and tungstates . In: Acta Cryst. B26, 1970, p. 19741-1977 , doi : 10.1107 / S0567740870005277 .
- ↑ a b c d e f g data sheet cesium tungstate from AlfaAesar, accessed on June 12, 2016 ( PDF )(JavaScript required) .
- ↑ a b Jane E. Macintyre: Dictionary of Inorganic Compounds . CRC Press, 1992, ISBN 978-0-412-30120-9 , pp. 3097 ( limited preview in Google Book Search).

