Dibromotoluenes
Dibromotoluenes (also dibromotoluenes or dibromomethylbenzenes ) form a group of substances that is derived from both toluene and bromobenzene . The structure consists of a benzene ring with an attached methyl group (–CH 3 ) and two bromine atoms (–Br) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 7 H 6 Br 2 . Dibromotoluenes therefore belong to the group of multiply brominated toluenes.
properties
The dibromotoluenes are practically insoluble in water; however, they are with other organic solvents , such as. B. benzene , alcohols , ethers , ethyl acetate and bromoform , soluble.
| Dibromotoluenes | ||||||||||||
| Surname | 2,3-dibromotoluene | 2,4-dibromotoluene | 2,5-dibromotoluene | 2,6-dibromotoluene | 3,4-dibromotoluene | 3,5-dibromotoluene | ||||||
| other names | 1,2-dibromo- 3-methylbenzene |
1,3-dibromo- 4-methylbenzene |
1,4-dibromo- 3-methylbenzene |
1,3-dibromo- 2-methylbenzene |
1,2-dibromo- 4-methylbenzene |
1,3-dibromo- 5-methylbenzene |
||||||
| Structural formula |

|

|

|

|

|
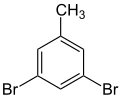
|
||||||
| CAS number | 61563-25-5 | 31543-75-6 | 615-59-8 | 69321-60-4 | 60956-23-2 | 1611-92-3 | ||||||
| ECHA InfoCard | 100,046,066 | 100.009.489 | 100.125.418 | 100.110.682 | ||||||||
| PubChem | 34680 | 94421 | 12006 | 34681 | 34679 | 15361 | ||||||
| Molecular formula | C 7 H 6 Br 2 | |||||||||||
| Molar mass | 249.93 g mol −1 | |||||||||||
| Physical state | liquid | firmly | ||||||||||
| Brief description | colorless liquid | colorless liquid | light yellow solid | |||||||||
| Melting point | −10 ° C | 5-6 ° C | 2-6 ° C | −10 ° C | 34-38 ° C | |||||||
| boiling point | 243 ° C | 135-136 ° C (35 mmHg) | 112-113 ° C (7 mmHg) | 91-92 ° C at 1,013 hPa | 246 ° C | |||||||
| density | 1.85 g cm −3 | 1.815 g cm −3 | 1.812 g cm −3 | 1.807 g cm −3 | ||||||||
| Refractive index | 1.601 | 1.602 | 1.606 | 1,600 | ||||||||
| solubility | practically insoluble in water | |||||||||||
|
GHS labeling |
|
|
|
|
|
|
||||||
| H and P phrases | see above | 315-319-335 | 315-319-335 | 315-319 | 315-319-335 | 315-319-335 | ||||||
| see above |
261-280-305 + 351 + 338 304 + 340-405-501 |
261-305 + 351 + 338 | 280-305 + 351 + 338 | 261-305 + 351 + 338 | 261-305 + 351 + 338 | |||||||
Extraction and presentation
The dibromotoluenes can be obtained by bromination of bromotoluenes , which in turn are produced from the toluidines by means of the Sandmeyer reaction .
See also
- Bromotoluenes
- Bromobenzyl bromides
- Tribromotoluenes , C 7 H 5 Br 3
- Tetrabromotoluenes , C 7 H 4 Br 4
- Pentabromotoluene , C 7 H 3 Br 5
Individual evidence
- ↑ a b c d e f data sheet 2,5-dibromotoluene, 98% from Sigma-Aldrich , accessed on June 8, 2015 ( PDF ).
- ↑ a b c d e f data sheet 2,6-dibromotoluene, 98 +% from AlfaAesar, accessed on June 8, 2015 ( PDF )(JavaScript required) .
- ↑ a b c data sheet 3,5-dibromotoluene, 97% from Sigma-Aldrich , accessed on June 8, 2015 ( PDF ).
- ↑ a b c d e data sheet 2,4-dibromotoluene, 98% from AlfaAesar, accessed on June 8, 2015 ( PDF )(JavaScript required) .
- ↑ a b c d e data sheet 3,4-dibromotoluene, 97% from Sigma-Aldrich , accessed on June 8, 2015 ( PDF ).
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 96th Edition . CRC Press, 2015, ISBN 978-1-4822-6097-7 , pp. 152 ( limited preview in Google Book search).
- ^ Association of authors: Organikum , 19th edition, Johann Ambrosius Barth, Leipzig · Berlin · Heidelberg 1993, ISBN 3-335-00343-8 , p. 566.
