Dichlorophenols
The dichlorophenols ( DCP ) form a group of aromatic compounds from the group of chlorophenols , which are derived from phenol as well as from chlorobenzene or dichlorobenzenes . The structure consists of a benzene ring with an attached hydroxyl group (-OH) and two chlorine atoms (-Cl) as substituents . Their different arrangement results in six constitutional isomers . They have the empirical formula C 6 H 4 Cl 2 O, a molar mass of 163.00 g / mol and are in the form of colorless crystal needles. During their production, polychlorinated dibenzodioxins and dibenzofurans are produced as by-products under unfavorable conditions . In particular, 2,4-dichlorophenol is used in the manufacture of herbicides .
properties
| Dichlorophenols | |||||||||||||||||||
| Surname | 2,3-dichlorophenol, 1,2-dichloro- 3-hydroxybenzene |
2,4-dichlorophenol , 1-hydroxy- 2,4-dichlorobenzene |
2,5-dichlorophenol, 1,4-dichloro- 2-hydroxybenzene |
2,6-dichlorophenol, 1,3-dichloro- 2-hydroxybenzene |
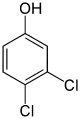
3,4-dichlorophenol, 1,2-dichloro- 4-hydroxybenzene |
3,5-dichlorophenol, 1,3-dichloro- 5-hydroxybenzene |
|||||||||||||
| Structural formula |

|

|

|

|
|

|
|||||||||||||
| CAS number | 576-24-9 | 120-83-2 | 583-78-8 | 87-65-0 | 95-77-2 | 591-35-5 | |||||||||||||
| 25167-81-1 (mixture of isomers) | |||||||||||||||||||
| PubChem | 11334 | 8449 | 66 | 6899 | 7258 | 11571 | |||||||||||||
| ECHA InfoCard | 100.008.546 | 100.004.027 | 100.008.657 | 100.001.602 | 100.002.228 | 100.008.833 | |||||||||||||
| 100.042.433 (mixture of isomers) | |||||||||||||||||||
| Molecular formula | C 6 H 4 Cl 2 O | ||||||||||||||||||
| Molar mass | 163.00 g mol −1 | ||||||||||||||||||
| Physical state | firmly | ||||||||||||||||||
| Brief description | flammable, colorless to yellowish crystal needles with a phenol-like odor | ||||||||||||||||||
| Melting point | 56-57 ° C | 40-43 ° C | 56-58 ° C | 64-66 ° C | 68 ° C | 65-67 ° C | |||||||||||||
| boiling point | 206 ° C | 209-211 ° C | 211 ° C | 218-220 ° C | 253 ° C | 233 ° C | |||||||||||||
| solubility | poorly soluble in water | ||||||||||||||||||
|
GHS labeling |
|
|
|
|
|
|
|||||||||||||
| H and P phrases | 319-302 + 312-315-411 | 302-311-314-411 | 302-315-319-335-411 | 314 | 302-315-318 | 302-311-314-411 | |||||||||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | ||||||||||||||
|
280-302 + 352 305 + 351 + 338-273-501 |
273-280-301 + 330 + 331 302 + 350-305 + 351 + 338-310 |
273-280-301 + 312 302 + 352-305 + 351 + 338 |
280-302 + 352-305 + 351 + 338 | 280-305 + 351 + 338 | 273-280-305 + 351 + 338-310 | ||||||||||||||
| LD 50 | 2376 mg kg −1 oral, mouse |
47 mg kg −1 oral, rat |
580 mg kg −1 oral, rat |
- | 1685 mg kg −1 oral, mouse |
2389 mg kg −1 oral, mouse |
|||||||||||||
use
2,4-dichlorophenol is used in moth repellants and as an antifungal agent in stockings, shoe insoles, etc. as well as in the manufacture of herbicides such as 2,4-dichlorophenoxyacetic acid , which made up half of the defoliant Agent Orange used in the Vietnam War . 3,5-dichlorophenol is used as a reference substance in some standardized bio-test procedures for water, such as the luminescent bacteria test.
Biological importance
2,6-dichlorophenol is used as a pheromone by the females of 14 tick species and 2,4-dichlorophenol is produced as a growth hormone by penicillin molds .
Individual evidence
- ↑ a b Entry on dichlorophenol, isomers in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d Entry on 2,3-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d Entry on 2,4-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d Entry on 2,5-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d Entry on 2,6-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c Entry on 3,4-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d Entry on 3,5-dichlorophenol in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ Data sheet 3,4-dichlorophenol from Sigma-Aldrich , accessed on December 27, 2019 ( PDF ).
- ↑ a b c d e f data sheet at chemicalland21.com
- ↑ Gordon W. Gribble: Environmental poisons from the gift table of nature . In: Spectrum of Science 06/2005.



