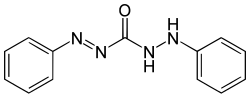
Diphenylcarbazone
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Diphenylcarbazone | ||||||||||||||||||
| other names |
1,5-diphenylcarbazone |
||||||||||||||||||
| Molecular formula | C 13 H 12 N 4 O | ||||||||||||||||||
| Brief description |
odorless, yellow to red solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 240.26 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
157 ° C |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Diphenylcarbazon is a chemical compound from the group of carbazones (nitrogen compounds with the basic structure HN = N-CO-NH-NH 2 ).
properties
Diphenylcarbazone is an orange solid that dissolves well in ethanol , not water. It forms a purple complex compound with Hg (II) ions . Likewise, other metal ions, such as. B. Cr (III) ions, colored complexes. Diphenylcarbazone can be produced from diphenylcarbazide via oxidation . Certain commercial products of diphenylcarbazone are a mixture with diphenylcarbazide, which also forms colored complex compounds with certain metal ions.
use
Diphenylcarbazone is used as an indicator to determine the end point in mercurimetry:
If a sodium chloride solution is titrated with mercury (II) nitrate solution , undissociated mercury (II) chloride is formed . When the end point is exceeded, the color complex is then formed with diphenylcarbazone. This method is used in water analysis for the determination of chloride.
If a certain amount of mercury (II) nitrate solution is added in excess to a chloride solution, the color intensity of the complex can be determined photometrically and thus the chloride content can be concluded.
literature
- K. Höll: Wasser, Verlag de Gruyter Berlin, 6th edition 1979, p. 55, ISBN 3-11-007560-1 .
- Lange / Zdenĕk: Photometric analysis, Verlag Chemie Weinheim 1980, p. 340, ISBN 3-527-25853-1 .
Related links
Individual evidence
- ↑ a b c d e Entry on diphenylcarbazone at TCI Europe, accessed on June 27, 2011.
- ↑ a b Entry on 1,5-diphenylcarbazone. In: Römpp Online . Georg Thieme Verlag, accessed on August 1, 2014.
