HBcAg
| HBcAg | ||
|---|---|---|
| other names |
Core antigen, capsid protein |
|
| Mass / length primary structure | 185 amino acids , 21,332 Da | |
| Identifier | ||
| External IDs | ||
| Orthologue ( hepatitis B virus ) | ||
| Entrez | 944568 | |
| UniProt |
P03148
|
|
| PubMed search |
944568
|
|
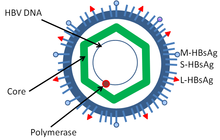
HBcAg ( hepatitis B core antigen ) is a protein of the hepatitis B virus (HBV).
properties
HBcAg is the capsid protein of HBV. It forms multimers up to an icosahedral capsid of 240 HBcAg with a triangulation number T = 4 and 180 HBcAg with T = 3. After unpacking, the HBcAg is transported in the cell along microtubules into the nucleus . A phosphorylation of the HBcAg exposes a signal sequence near the C -terminus (position 187-204) for import into the nucleus through the importin α and β. The capsid binds to NUP153 in the cell nucleus . This is where the HBV genomes are unpacked, provided they have fully matured. However, various incomplete viral and virus-like particles are formed.
The capsid domain is at position 1 to 144, while from 150 to 185 there is an arginine- rich, protamine- like nucleic acid-binding domain.
HBeAg is formed from the same open reading frame as HBcAg. Newly formed HBcAg in the cytosol binds pre-genomic RNA (at HBcAg position 206–212) and the polymerase protein P , whereupon the DNA genome of the HBV is generated from the RNA template. The N terminus contains four cysteines that are believed to form a zinc finger . Then either a new import into the cell nucleus takes place or the capsid binds to the endoplasmic reticulum for budding .
Tapasin binds to the section 18-27 of HBcAg and increases the activity of cytotoxic T cells against HBV.
Applications
HBcAg is used to detect current infection. After a single infection, the HBc antigen always remains positive during the infection. A person who has been vaccinated against HBV can be differentiated from someone who is currently or previously sick with HBV.
It is also being studied as a target for treating hepatitis B infections .
literature
- B. Böttcher, SA Wynne, RA Crowther: Determination of the fold of the core protein of hepatitis B virus by electron cryomicroscopy. In: Nature. Volume 386, Number 6620, March 1997, pp. 88-91, doi : 10.1038 / 386088a0 , PMID 9052786 .
- A. Zlotnick, B. Venkatakrishnan, Z. Tan, E. Lewellyn, W. Turner, S. Francis: Core protein: A pleiotropic keystone in the HBV lifecycle. In: Antiviral research. Volume 121, September 2015, pp. 82-93, doi : 10.1016 / j.antiviral.2015.06.020 , PMID 26129969 , PMC 4537649 (free full text).
Web links
Individual evidence
- ↑ a b c d e f g Uniprot : C - Capsid protein - Hepatitis B virus genotype A2 subtype adw2 (strain Rutter 1979) (HBV-A) - C gene & protein. In: uniprot.org. June 20, 2018, accessed February 26, 2018 .
- ^ J. Hu, K. Liu: Complete and Incomplete Hepatitis B Virus Particles: Formation, Function, and Application. In: Viruses. Volume 9, number 3, 03 2017, p., Doi : 10.3390 / v9030056 , PMID 28335554 , PMC 5371811 (free full text).
- ↑ M. Seifer, DN Standring: A protease-sensitive hinge linking the two domains of the hepatitis B virus core protein is exposed on the viral capsid surface. In: Journal of virology. Volume 68, Number 9, September 1994, pp. 5548-5555, PMID 7520091 , PMC 236955 (free full text).
- ↑ Pfam : pfam08290 Hep_core_N: Hepatitis core protein, putative zinc finger .
- Jump up ↑ X. Chen, Y. Tang, Y. Zhang, M. Zhuo, Z. Tang, Y. Yu, G. Zang: Tapasin modification on the intracellular epitope HBcAg18-27 enhances HBV-specific CTL immune response and inhibits hepatitis B virus replication in vivo. In: Laboratory investigation; a journal of technical methods and pathology. Volume 94, number 5, May 2014, pp. 478-490, doi : 10.1038 / labinvest.2014.6 , PMID 24614195 .
- ↑ T. Kimura, A. Rokuhara, A. Matsumoto, S. Yagi, E. Tanaka, K. Kiyosawa, N. Maki: New enzyme immunoassay for detection of hepatitis B virus core antigen (HBcAg) and relation between levels of HBcAg and HBV DNA. In: Journal of clinical microbiology. Volume 41, Number 5, May 2003, pp. 1901-1906. ISSN 0095-1137 , PMID 12734224 , PMC 154683 (free full text).
- ↑ T. Cao, P. Meuleman, I. Desombere, M. Sällberg, G. Leroux-Roels: In vivo inhibition of anti-hepatitis B virus core antigen (HBcAg) immunoglobulin G production by HBcAg-specific CD4 (+) Th1- type T-cell clones in a hu-PBL-NOD / SCID mouse model. In: Journal of virology. Volume 75, Number 23, December 2001, pp. 11449-11456. ISSN 0022-538X , doi: 10.1128 / JVI.75.23.11449-11456.2001 , PMID 11689626 , PMC 114731 (free full text).
- ↑ LY Mak, DK Wong, WK Seto, CL Lai, MF Yuen: Hepatitis B core protein as a therapeutic target. In: Expert opinion on therapeutic targets. Volume 21, number 12, 12 2017, pp. 1153-1159, doi : 10.1080 / 14728222.2017.1397134 , PMID 29065733 .