Endoplasmic reticulum
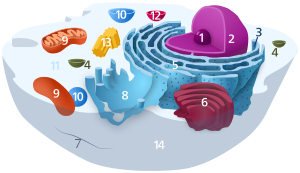
1. Nucleolus (nuclear body)
2. Cell nucleus (nucleus)
3. Ribosomes
4. Vesicle
5. Rough (granular) ER (ergastoplasm)
6. Golgi apparatus
7. Cytoskeleton
8. Smooth (agranular) ER
9 . mitochondria
10. lysosome
11. cytoplasm (with cytosol and cytoskeleton )
12. peroxisomes
13. centrioles
14 cell membrane

| Parent |
| Organelle |
| Subordinate |
| ER membrane ER lumen ribosome protein complexes |
| Gene Ontology |
|---|
| QuickGO |
The endoplasmic reticulum ( ER , endoplasmic "in the cytoplasm ", lat. Reticulum "throwing net") is a branched canal system of flat cavities, which is enclosed by membranes . The endoplasmic reticulum is found in all eukaryotic cells with the exception of mature erythrocytes ; depending on the cell type, it is developed to different degrees.
construction
The endoplasmic reticulum consists of a widely branched membrane network of tubes, vesicles and cisterns (sac-like structures) that are surrounded by the ER membrane. The ER membrane closes the interior of the endoplasmic reticulum, the ER lumen , from the cytosol . The membrane labyrinth of the endoplasmic reticulum makes up over half of the total amount of membrane in a eukaryotic cell.
The ER membrane merges directly into the nuclear envelope of the cell nucleus, that is, the nuclear envelope and ER represent a morphological continuum. The ER lumen is connected to the membrane space between the nuclear envelope, the perinuclear space .
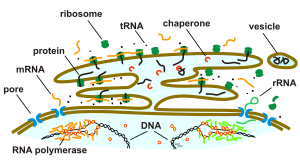
Parts of the endoplasmic reticulum, called the rough ER , are covered with ribosomes on their membrane surfaces ; other areas are smooth and ribosome-free and are therefore called smooth ER . Rough and smooth ER differ in their function.
The structure of the endoplasmic reticulum is dynamic and subject to constant reorganization. This includes the elongation or retraction of membrane tubules, their branching, fusion or splitting. This ER motility depends on the cytoskeleton . In plant cells and yeast, F-actin in particular plays an important role. In animal cells, on the other hand, the ER is built up and remodeled under the dominant influence of the microtubules . It was shown that a representative of the actin-associated motor proteins of the myosin family myosin V is responsible for the transmission of the peripheral ER to the daughter cells during cell division.
Tasks and types
Translation , protein folding , protein quality control , post-translational modifications of proteins and protein transport of transmembrane proteins and secretory proteins (see exocytosis ) take place on and in the ER . In addition, the ER is the place where (e.g. after mitosis ) new nuclear membranes are formed and constricted. The ER also serves as an intracellular calcium store, which means that it plays a key role in signal transduction . In muscle cells (where the ER is called sarcoplasmic reticulum , SR for short), the release of calcium is the mediator of a contraction. The tasks of rough and smooth ER and SR are different.
Smooth ER (agranular ER)
The smooth ER ( English smooth endoplasmic reticulum , sER) plays an important role in several metabolic processes (= metabolic processes). Smooth ER enzymes are important for the synthesis of various lipids (especially phospholipids , fatty acids and steroids ). The smooth ER also plays an important role in carbohydrate metabolism, detoxification of the cell and the storage of calcium. Accordingly, the ER found in parenchymal cells of the kidney and liver is predominantly smooth ER.
Hormone synthesis
The steroids formed in the smooth ER include steroid hormones , such as those of the adrenal cortex or the sex hormones of vertebrates. For example, the cells in the testes and ovaries that are responsible for producing hormones contain a particularly large amount of smooth ER.
Carbohydrate storage
Glucose is stored in the liver's parenchymal cells (= hepatocytes ) as a polymer called glycogen . The release of single molecules (= monomers ) from glycogen is called glycogenolysis . Glycogenolysis in liver cells is an important process for controlling the glucose concentration in the blood (= the blood sugar level ). Glycogenolysis does not produce glucose itself, but glucose-6-phosphate . The enzyme glucose-6-phosphatase on the membrane of the smooth ER splits off the phosphate group of glucose-6-phosphate; (Only) the resulting glucose can leave the liver cell and thus increase the blood sugar level.
detoxification
The smooth ER of the hepatocytes also contains membrane-bound enzymes that are involved in the biotransformation of exogenous substances (e.g. drugs). These enzymes (so-called. CYP s) belong to the Cy tochrom- P 450 class , and also in some other tissues outside the liver expressed . In the liver, CYPs have a detoxification function; they can recognize a large number of exogenous substances as substrates . CYPs mostly oxidize their substrates and thereby insert polar groups into them. The resulting substances are more water-soluble than the exogenous starting substances and can therefore be excreted more easily by the kidneys (= are kidney-accessible).
Calcium storage
In the lumen of the ER, the calcium 2+ concentration reaches millimolar values (approx. 10 −3 M). In the cytosol , on the other hand, the concentration of free calcium ions at rest is only around 100–150 n M (i.e. around 10 −7 M). Thus there is a concentration gradient of four orders of magnitude across the membrane of the ER . Both the uptake of calcium in the ER and the release of calcium ions from the ER are subject to fine regulation under physiological conditions, which is extremely important for maintaining calcium homeostasis .
Since calcium ions in the cytosol are an important “ second messenger ”, the regulated release of calcium from the ER plays a key role in intracellular signaling. The effects of an increase in the intracellular calcium concentration due to release from the ER are diverse:
- Enzymes are activated or inhibited,
- the gene expression is regulated,
- synaptic plasticity is influenced in neurons ,
- The muscle fibers contract in the muscles (calcium ions are released from the ER, which in muscle cells is called the sarcoplasmic reticulum (SR)),
- Immune system cells release antibodies , etc.
Calcium ions leave the ER through two types of calcium channels : the IP 3 receptors and the ryanodine receptors . The abbreviation IP 3 stands for inositol trisphosphate , which is also a second messenger . IP 3 arises as a cleavage product of phosphatidylinositol-4,5-bisphosphate through the mediation of phospholipase C , which in turn is activated by certain G proteins (Gq). This happens when a metabotropic receptor coupled to this G protein is excited in the plasma membrane . - The IP 3 generated in this way binds to its specific receptor in the membrane of the ER, whereupon the calcium concentration in the cytoplasm increases due to the outflow from the ER through the channels of the IP 3 receptors. As part of this signal chain (metabotropic receptor - G protein - phospholipase C - IP 3 - IP 3 receptor - calcium release) calcium can also be viewed as a tertiary messenger.
The ryanodine receptors are calcium-sensitive calcium channels. So on the one hand they are permeable to calcium and on the other hand they are activated by calcium ions. This happens when the calcium concentration in the cytoplasm increases. Calcium ions bind to the ryanodine receptors, these open, and calcium ions flow through them from the ER into the cytosol. This process is called " calcium-induced calcium release (" English. " CICR " - calcium-induced calcium release ). Best known is the role of CICR in the contraction of myocardial cells .
Calcium ATPases of the SERCA type are located in the membrane of the ER . SERCA stands for Sarcoendoplasmic Reticulum ATPase . These proteins transport calcium ions from the cytoplasm back into the ER. Since the above-mentioned steep concentration gradient has to be overcome, this transport process can only take place with consumption of ATP. This transport process is therefore one of the primarily active transport processes.
Membrane production
The smooth ER allows its own membrane to grow and directs membrane components via transport vesicles to other parts of the inner membrane system , i.e. the Golgi apparatus, endosomes, lysosomes and also the plasma membrane. The phospholipids are also produced by the smooth ER, as enzymes of the ER membrane put them together from precursor molecules that are located in the cytosol and in the ER membrane.
Transitory function
Vesicles bud from some sections of the smooth endoplasmic reticulum, which then fuse with the cis side of the Golgi apparatus and thus transfer proteins for further processing (e.g. adding sugar side chains). This is part of the so-called "secretory pathway" because it mainly affects proteins that are later excreted from the cell into the extracellular space through exocytosis .
Rough ER (granular ER)
The rough ER (rER), also called granular ER or ergastoplasm , has two functions: protein biosynthesis and membrane production. It gets its name from the ribosomes that sit on its membrane surfaces. It is mainly found in the cells of exocrine glands and the liver as well as in nerve ( Nissl clods ) and embryonic cells . The rough ER can be made visible with basic dyes such as hematoxylin , cresyl violet or toluidine blue ( Nissl stain ).
Protein synthesis
Proteins are often excreted by specialized cells ( secretion ). These proteins are made by the ribosomes attached to the rough ER. One of these proteins is, for example, the insulin from cells in the pancreas .
All polypeptide chains that develop in membrane-bound ribosomes are first channeled into the lumen of the ER. This happens through pore-forming proteins ( cotranslation ). Proteins synthesized in the cytosol are also transported into the lumen of the ER ( post-translation ). The polypeptide chains are cut to size and folded in the lumen of the ER.
After the translocation, the linear amino acid chains are folded into the ER, thus retaining their three-dimensional structure. This process is supported and controlled by other proteins in the ER ( chaperones ). Misfolded proteins are immediately retranslocated, that is, transported back into the cytosol and there degraded by the proteasome . The cholera bacterium uses this mechanism to bring its toxin into the cytosol via this process, where it escapes degradation by the proteasome and can develop its toxic effect.
Most secretion proteins are glycoproteins that carry covalently bound carbohydrates. These carbohydrates, they are oligosaccharides , are attached in the lumen of the ER by the enzymes of the ER. The finished secretory proteins remain in the lumen of the ER and are thus kept away from proteins in the cytosol, which were created by free ribosomes. The secretory proteins are pinched off in the form of small membrane vesicles and thus leave the lumen of the ER as a transport vesicle in the direction of the Golgi apparatus .
In 1999 Günter Blobel received the Nobel Prize for Physiology or Medicine for his discovery, made in 1975, that endogenous protein signals ( signal sequences ) are used to target proteins from the ER to various cell compartments . A certain N-terminal peptide sequence is referred to as a signal sequence in this sense, which is cleaved by the signal peptidase after transport through the membrane of the ER . Proteins intended for targets outside of the ER are then packaged in transport vesicles and transported along the cytoskeleton to their destination.
Sarcoplasmic Reticulum (Smooth Reticulum, SR)
The smooth ER in muscle cells is called the sarcoplasmic reticulum (SR). The SR is a specialized ER of the muscle cells. It stores calcium ions. These are released when an electrical impulse ( action potential ) arrives in the sarcoplasm (cytoplasm of the muscle cells), diffuse between the actin and myosin filaments of the muscle fibrils and cause the filaments to slide into one another. This causes the muscle fiber to contract. If no further excitations arrive at the muscle fiber, the calcium ions are actively pumped back into the SR. This prevents a renewed contraction. The sarcoplasmic reticulum is used to regulate muscle contraction . The sarcoplasmic reticulum is also part of the longitudinal tubular system.
literature
- Bruce Alberts et al: Molecular Biology of the Cell. 4th edition. Garland Science, New York 2002, ISBN 0-8153-4072-9 .
- Neil A. Campbell et al: biology. 1st edition, 1st corrected reprint Spectrum, Heidelberg 1997, ISBN 3-8274-0032-5 .
Web links
- Endoplasmic Reticulum (animation about the structural change of the ER)
- Rough ER: electron microscopic images (Dr. Jastrow's EM atlas)
- Smooth ER: electron microscopic images (Dr. Jastrow's EM atlas)
Individual evidence
- ↑ Wissenschaft-Online-Lexika: Entry on Ergastoplasma in the Lexicon of Biology, accessed on November 26, 2011.
- ^ Ergastoplasma in the Roche Medical Lexicon, 5th edition.