Cytoskeleton
| Parent |
| Organelle |
| Subordinate |
|
Microfilaments Intermediate Filaments Microtubules |
| Gene Ontology |
|---|
| QuickGO |

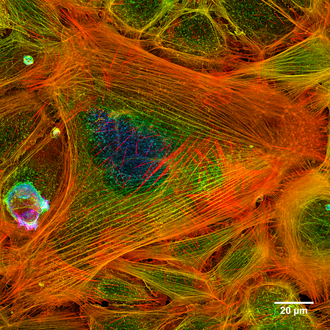
The cytoskeleton ( ancient Greek κύτος kýtos - "cell") (also cytoskeleton or cell skeleton ) is a network in the cytoplasm of eukaryotic cells made up of proteins . It consists of thin, thread-like cell structures ( microfilaments ) that can be dynamically built up and broken down . It is responsible for the mechanical stabilization of the cell and its external shape, for active movements of the cell as a whole, and for movements and transports within the cell.
The cytoskeleton may actually look like a skeleton in fluorescence microscopic images , but the name is misleading because the cytoskeleton is not a rigid skeleton or scaffolding, but a dynamic network of structures. It is now also known that cytoskeletal elements are essential not only for mechanical stability, but also for sensory functions such as signal transmission between cells.
The word cytoskeleton was coined in 1931 by the embryologist Paul Wintrebert , who speculated about how egg cells might manage to maintain their internal organization when they have to wander through the narrow female reproductive tract. From the strong deformation forces acting on the egg cell, Wintrebert derives the necessity of the existence of a cytoskeleton (“cytosquelette”).
The eukaryotic cytoskeleton

In the eukaryotic cell, a distinction is made between three classes of cytoskeletal filaments, which are each formed by different proteins or protein classes, have specific accompanying proteins and each participate in the tasks of the cytoskeleton in different ways:
All three classes are involved in the mechanical stabilization of the cell. Surface differentiations are supported by actin filaments and microtubules. All forms of active movement also take place along these two filament types, as they have specific motor proteins .
In general, the structures of the cytoskeleton are associated with accompanying proteins (including adapter proteins and motor proteins ) specific to the task and protein type, which stabilize the filaments, move on them or connect them to other structures (see for example Profilin ). The build-up and breakdown of the cytoskeleton can be inhibited by cytoskeleton inhibitors .
Microtubules
The most noticeable components of the cytoskeleton are the microtubules , hollow cylinders with a diameter of 25 nm, which are made up of the protein tubulin . Intracellularly, with their motor proteins dynein and kinesin, they are responsible for longer transport processes and the movements and attachment of the organelles in the cytosol. In the case of the mitotic spindle , the replicated chromosomes are drawn to the two nuclear poles. Microtubules only play a minor role in mechanical stabilization, but they are the characteristic internal framework of the mobile kinocilia .
The assembly and dismantling of the microtubules can be carried out very dynamically and in animal cells starts from the centrosome .
Actin filaments
Actin filaments (also called microfilaments) are fibers with a diameter of 7 nm that are made of actin . Especially in net-like arrangements below the plasma membrane and in membrane bulges ( microvilli , pseudopodia ) they stabilize the outer shape of the cell, keep membrane-bound proteins in place and move into certain cell functions ( adherence contact ). They can also be built up and taken down dynamically.
The motor proteins of actin form the protein class of myosins . Not only is the movement of the muscles based on the actin-myosin interaction , but myosins also tighten the actin filaments for stabilization and ensure short-distance transport, for example from vesicles to the plasma membrane (while long-distance transport is taken over by microtubules / dynein and kinesin).
Intermediate filaments
The term intermediate filaments covers a number of protein filaments, all of which have very similar properties. Their diameter is around 10 nm (8 to 11 nm), and because they are significantly more stable than microtubules and actin filaments, they can best absorb mechanical tensile forces. For this reason, they mainly serve to mechanically stabilize the cells. They form their supporting structure and radiate into certain cell connections ( desmosomes , hemidesmosomes ).
The prokaryotic cytoskeleton
Also prokaryotic cells, as have proteins homologous all three eukaryotic protein classes are considered to proteins. Even if they form similar structures, the amino acid sequences show hardly any similarities to the eukaryotic protein classes.

B - Model comparison of bacterial (blue) and eukaryotic (gray) microtubules. The eukaryotes include various unicellular organisms as well as multicellular animals, plants and fungi.
While eukaryotes , regardless of whether they are single-cell or multicellular such as plants, fungi or animals, have a fairly uniform tubulin (with a genetic similarity of 90-95%), prokaryotes have considerably more different tubulin-like proteins ( BtubA and BtubB , TubZ , RepX , FtsZ ), some of which only match 40% in their gene sequence. Nevertheless, the bacterial microtubules made up of these homologues seem to fulfill essentially the same functions as those of the eukaryotes.
FtsZ was found as the tubulin homologue and FtsA as the actin homologue . These proteins are particularly involved in cell division processes. In plants, FtsZ proteins are encoded by a small family of genes in the nucleus . They are mostly imported into the chloroplasts , where they contribute to the division of these cell organelles . From experiments on the deciduous moss Physcomitrella patens it is known that FtsZ proteins can also form a complex network in the chloroplasts. Since this network is strongly reminiscent of the cytoskeleton, Ralf Reski coined the term plastoskeleton for this structure in 2000 and postulated that it fulfills similar complex functions in the plastids as the cytoskeleton does for the entire cell. It was also in the bacterium Caulobacter crescentus the Crescentin found that resembles the intermediate filaments in its function.
Web links
- University of Leipzig: Cytoskeleton (English) ( Memento from April 2, 2009 in the Internet Archive )
- Zytologie-online.net: Information about the cytoskeleton of cells
Individual evidence
- ↑ F Huber, J Schnauss, S Roenicke, P Rauch, K Mueller, C Fuetterer, J Kaes: Emergent complexity of the cytoskeleton: from single filaments to tissue . In: Advances in Physics . 62, No. 1, 2013, pp. 1–112. doi : 10.1080 / 00018732.2013.771509 .
- ↑ P. Wintrebert: La rotation immédiate de l'oeuf pondu et la rotation d'activation chez Discoglossus pictus Otth. In: Comptes rendus des séances de la Société de biologie et de ses filiales. Vol. 106, 1931, ISSN 1295-0661 , pp. 439-442.
- ↑ Satyajit Sahu, Subrata Ghosh, Daisuke Fujita, Anirban Bandyopadhyay: Live visualizations of single isolated tubulin protein self-assembly via tunneling current: effect of electromagnetic pumping during spontaneous growth of microtubule. In: Scientific Reports. Volume 4, No. 7303, 2014.
- ↑ KK Busiek, W. Margolin: Bacterial actin and tubulin homologs in cell growth and division. In: Current Biology . Volume 25, number 6, March 2015, pp. R243 – R254, doi : 10.1016 / j.cub.2015.01.030 , PMID 25784047 , PMC 5519336 (free full text) (review).
- ↑ Martin Pilhofer, Mark S. Ladinsky, Alasdair W. McDowall, Giulio Petroni, Grant J. Jensen: Microtubules in Bacteria: Ancient Tubulins Build a Five-Protofilament Homolog of the Eukaryotic Cytoskeleton. In: Journal: PLOS Biology, Published: December 6, 2011, doi : 10.1371 / journal.pbio.1001213
- ^ Contzen Pereira: Cytoskeleton and Consciousness: An Evolutionary Based Review. In: Journal: NeuroQuantology, June 2015, Volume 13, Issue 2, Page 232-239
- ↑ Ralf Reski: Rings and networks: the amazing complexity of FtsZ in chloroplasts. In: Trends in Plant Science. Vol. 7, 2002, ISSN 1360-1385 , pp. 103-105, doi: 10.1016 / S1360-1385 (02) 02232-X .
- ^ Justine Kiessling, Sven Kruse, Stefan A. Rensing, Klaus Harter, Eva L. Decker, Ralf Reski: Visualization of a Cytoskeleton-like Ftsz Network in Chloroplasts. In: Journal of Cell Biology . Vol. 151, No. 4, November 13, 2000, ISSN 0021-9525 , pp. 945-950, online .
- ^ Geoffrey I. McFaddena: Skeleton in the closet. How do chloroplasts stay in shape? In: The Journal of Cell Biology. Vol. 151, No. 4, November 13, 2000, F19-F21, online .