Medroxyprogesterone
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
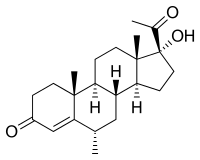
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Medroxyprogesterone | |||||||||||||||||||||
| other names |
17 α - hydroxy -6 α -methylpregn-4-en-3,20-dione ( IUPAC ) |
|||||||||||||||||||||
| Molecular formula | C 22 H 32 O 3 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 344.49 g · mol -1 | |||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Medroxyprogesterone or Medroxyprogesterone Acetate (MPA) is a synthetic progestin that is used as an ovulation inhibitor for contraception in the three-month injection .
application
- Ovulation inhibition: (suppression of the normal LH peak before ovulation) as an intramuscular injection (3-month injection) with 150 mg / 2 ml (as Depo-Provera)
- Disturbance of follicle maturation
- Disruption of the proliferation of progesterone-sensitive tissues: endometrium , breast: for the treatment of endometrial cancer , breast cancer with 400 mg to 1000 mg / week initially, 400 mg / month thereafter, and endometriosis with 3 × 10 mg / day for 90 days with day-1 of the cycle
- Prostate cancer , initially with 500 mg, 2 times / week for 3 months
- Increase in the viscosity of the cervical mucus (barrier for sperm), also approved in the UK in the treatment of renal cell carcinoma
- Hormone replacement therapy together with estrogens (10 mg / day for the last 14 days of the cycle)
safety instructions
The No Observed Effect Level ( NOEL ) of medroxyprogesterone is 30 μg / day / kg body weight, the Acceptable Daily Intake ( ADI ) is 18 μg / day / kg body weight.
Use in animals
In Germany it is not approved for use in food-producing animals. A health assessment of possible residues in food after zootechnical use in sheep by the EU led to the result that no maximum amount has to be set for MPA due to the safety, if the use is intravaginal and only for zootechnical purposes.
In dogs and cats, the active ingredient may be, for termination of the heat or heat cycle and for the prevention of pregnancy may be used.
Other Information
In the summer of 2002, it became known that a pharmaceutical company had transferred industrial waste containing medroxyprogesterone acetate to an environmental technology company. A sugar manufacturer, in turn, had incorporated this material into animal feed over two years and also sold it to Germany. MPA residues were found in glucose syrup from the company concerned and in pork.
Trade names
Depocon (A), Depo-Clinovir (D), Depo-Provera (CH), Farlutal (A, CH), Prodafem (A, CH), Sayana (A), various generics (D)
Climopax (D), Indivina (D, CH), Osmil (D), Sisare (D), Triaval (CH)
- Veterinary medicine
Perlutex, Supprestral, Sedometril
Individual evidence
- ↑ a b Datasheet Medroxyprogesterone from Sigma-Aldrich , accessed on April 9, 2011 ( PDF ).
- ↑ External identifiers or database links for medroxyprogesterone acetate : CAS number: 71-58-9, EC number: 200-757-9, ECHA InfoCard: 100.000.689 , PubChem : 6279 , ChemSpider : 6043 , DrugBank : DB00603 , Wikidata : Q2823834 .
- ↑ British National Formula, September 2006, pp. 383, 464; bnf.org
- ↑ ( Page no longer available , search in web archives: Summary Report of the EMA ) (PDF; 41 kB).
- ^ Opinion of the BgVV of July 11, 2002 (PDF; 40 kB).
- ↑ Press release of the BMVEL (today BMELV) on PMA residues from July 25, 2002 (PDF; 20 kB).