N 4 , N 4 -dimethylcytosine
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
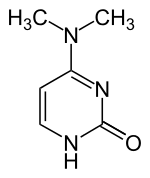
|
|||||||||||||
| General | |||||||||||||
| Surname | N 4 , N 4 -dimethylcytosine | ||||||||||||
| other names |
4-dimethylamino-pyrimidin-2-one |
||||||||||||
| Molecular formula | C 6 H 9 N 3 O | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 139.16 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
N 4 , N 4 -Dimethylcytosine is a heterocyclic organic compound with a pyrimidine backbone. It is a derivative of the nucleobase cytosine , which isdoubly methylated at the amino group . It occurs as a component of the nucleoside N 4 , N 4 -dimethylcytidine (m 4 2 C).
Further dimethylated nucleobases are N 2 , N 2 -dimethylguanine and N 6 , N 6 -dimethyladenine .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.