Promethium (III) oxide
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
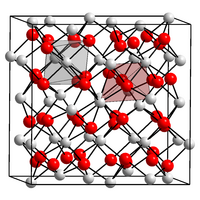
|
||||||||||
| __ Pm 3+ __ O 2− | ||||||||||
| Crystal system | ||||||||||
| Space group |
Ia 3 (No. 206) |
|||||||||
| Lattice parameters |
a = 1099 pm |
|||||||||
| General | ||||||||||
| Surname | Promethium (III) oxide | |||||||||
| Ratio formula | Pm 2 O 3 | |||||||||
| Brief description |
coral red solid (C type) |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 337.82 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| density |
6.85 g cm −3 |
|||||||||
| Melting point |
2130 ° C |
|||||||||
| Hazard and safety information | ||||||||||
 Radioactive |
||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Promethium (III) oxide (Pm 2 O 3 ) is the oxide of the rare earth metal promethium .
presentation
Promethium (III) oxide is produced from promethium (III) oxalate by annealing at 800 ° C.
properties
It comes in three modifications (A, B, and C types). The A-type has a hexagonal crystal lattice (violet-brown), the B-type (violet-pink) is a monoclinic distortion of the A-type and the C-type has an isomorphic- cubic manganese (III) oxide structure (coral red). At room temperature it crystallizes in the C-type, the other two structures are only stable at higher temperatures.
When heated to 750–800 ° C, the cubic form changes into the monoclinic form; this change can only be reversed by melting the oxide. The conversion from the monoclinic to the hexagonal form takes place at 1740 ° C. The melting point is 2130 ° C.
shape Space group Pearson symbol No. a, b, c (pm) β (deg) Z Density
(g / cm 3 )cubic Ia 3 (No. 206) cI80 206 1099 16 6.85 monoclinic C 2 / m (No. 12) mS30 12 1422; 365; 891 100.1 6th 7.48 hexagonal P 3 m 1 (No. 164) hP5 164 380.2; 380.2; 595.4 1 7.62
- * a, b and c are lattice parameters , Z is the number of formula units per unit cell , the density is calculated from X-ray structure data.
The specific magnetic susceptibility is 15.5 · 10 −6 cm 3 / g, the molar magnetic susceptibility χ mol is 2660 · 10 −6 cm 3 / g.
safety instructions
Classifications according to the CLP regulation are not available because these only include chemical hazard and play a completely subordinate role compared to the hazards based on radioactivity . The latter also only applies if the amount of substance involved is relevant.
literature
- Fritz Weigel: The chemistry of promethium , in: Fortschr. Chem. Forsch. , 1969 , 12 (4), pp. 539-621 ( doi : 10.1007 / BFb0051097 ).
- James E. Huheey: Inorganische Chemie , 1st edition, de Gruyter, Berlin 1988, ISBN 3-11-008163-6 , pp. 873-900.
Individual evidence
- ↑ a b c d e f Weigel: Die Chemie des Promethiums , pp. 591-594.
- ↑ a b T. D. Chikalla, CE McNeilly, FP Roberts: Polymorphic Modifications of Pm 2 O 3 , in: Journal of the American Ceramic Society , 1972 , 55 (8), p. 428 ( doi : 10.1111 / j.1151-2916.1972. tb11329.x ).
- ↑ a b Gmelin's Handbook of Inorganic Chemistry , System No. 39, Part C 1, pp. 312-313.
- ↑ The hazards emanating from radioactivity do not belong to the properties to be classified according to the GHS labeling. With regard to other hazards, this substance has either not yet been classified or a reliable and citable source has not yet been found.
- ^ JC Sheppard, EJ Wheelwright, FP Roberts: The Magnetic Susceptibility of Promethium-147 Oxide , in: J. Phys. Chem. , 1963 , 67 (7), pp. 1568-1569 ( doi : 10.1021 / j100801a510 ).