Purpurin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
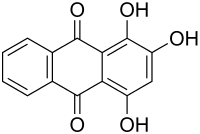
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Purpurin | ||||||||||||||||||
| other names |
1,2,4-trihydroxyanthraquinone |
||||||||||||||||||
| Molecular formula | C 14 H 8 O 5 | ||||||||||||||||||
| Brief description |
dark brown solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 256.21 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
253-256 ° C |
||||||||||||||||||
| solubility |
soluble in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Purpurin is a naturally occurring chemical compound and, like flavopurpurin and anthrapurpurin, belongs to the alizarin dyes . Purpurin occurs next to alizarin in the madder root . It is artificially made from alizarin by oxidation with manganese dioxide and sulfuric acid , but plays a minor role as a coloring agent (used in calico printing, chrome lacquer red-violet).
Individual evidence
- ↑ a b c d data sheet Purpurin from Sigma-Aldrich , accessed on October 22, 2016 ( PDF ).
- ↑ Purpurin data sheet at Acros, accessed February 20, 2010.
literature
- Paul Karrer: Textbook of Organic Chemistry . 10th edition, Georg Thieme Verlag, Stuttgart 1948, p. 630.
