Samarium (II) chloride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
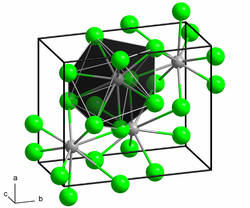
|
||||||||||||||||
| __ Sm 2+ __ Cl - | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Samarium (II) chloride | |||||||||||||||
| other names |
Samarium dichloride |
|||||||||||||||
| Ratio formula | SmCl 2 | |||||||||||||||
| Brief description |
dark brown solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 221.27 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
3.69 g cm −3 |
|||||||||||||||
| Melting point |
850 ° C |
|||||||||||||||
| boiling point |
1310 ° C |
|||||||||||||||
| solubility |
soluble in tetrahydrofuran |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Samarium (II) chloride is an inorganic chemical compound of samarium from the group of chlorides .
Extraction and presentation
Samarium (II) chloride can be obtained by reducing samarium (III) chloride with samarium in a vacuum at 800 to 900 ° C or hydrogen at 350 ° C, although the latter reaction requires long reaction times and undesirable side reactions occur.
The representation by reducing the samarium (III) chloride with zinc in a melt flow of zinc (II) chloride at 500 ° C is also possible.
The link can also by reduction with lithium - naphthalene win. Other reducing agents such as sodium or magnesium can also be used.
properties
Samarium (II) chloride is a dark brown solid. The connection is extremely hygroscopic and can only be stored and handled under carefully dried protective gas or in a high vacuum. In air or on contact with water, it changes into hydrates while absorbing moisture , but these are unstable and more or less quickly turn into oxide chlorides with evolution of hydrogen . The compound has a crystal structure of lead (II) chloride type. A cubic modification also exists above 680 ° C.
use
Samarium (II) chloride (as well as samarium (II) iodide and samarium (II) bromide ) in organic chemistry, for example pinacol - coupling reactions used.
Individual evidence
- ↑ a b c d e f g Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 1081.
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 93rd Edition . CRC Press, 2012, ISBN 1-4398-8049-2 , pp. 4–86 ( limited preview in Google Book Search).
- ↑ a b Jane E. Macintyre: Dictionary of Inorganic Compounds . CRC Press, 1992, ISBN 0-412-30120-2 , pp. 2903 ( limited preview in Google Book Search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Kurt Rossmanith: Production of the classic rare earth (II) chlorides in solution. In: Monthly magazine for chemistry. 110, 1979, pp. 109-114, doi : 10.1007 / BF00903752 .
- ↑ Advances in Inorganic Chemistry and Radiochemistry . Academic Press, 1977, ISBN 0-08-057869-1 , pp. 9 ( limited preview in Google Book search).
- ↑ David J. Procter, Robert A. Flowers, Troels Skrydstrup: Organic Synthesis Using Samarium Diiodide: A Practical Guide . Royal Society of Chemistry, 2010, ISBN 1-84755-110-6 , pp. 157 ( limited preview in Google Book search).


