Thulium (III) chloride
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||
| __ Tm 3+ __ Cl - | ||||||||||
| Space group |
C 2 / m (No. 12) |
|||||||||
| General | ||||||||||
| Surname | Thulium (III) chloride | |||||||||
| other names |
Thulium trichloride |
|||||||||
| Ratio formula | TmCl 3 | |||||||||
| Brief description |
light yellow powder |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | ||||||||||
| Physical state |
firmly |
|||||||||
| density |
4.34 g cm −3 |
|||||||||
| Melting point |
824 ° C |
|||||||||
| boiling point |
1490 ° C |
|||||||||
| solubility |
soluble in water |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| Toxicological data | ||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Thulium (III) chloride is a chemical compound from the group of chlorides .
Extraction and presentation
Thulium (III) chloride can be obtained by reacting thulium (III) oxide or thulium (III) carbonate and ammonium chloride.
The hexahydrate is formed by the reaction of thulium with hydrochloric acid . This can be converted to the anhydrate form by reaction with thionyl chloride .
Thulium (III) chloride can also be synthesized directly from the elements thulium and chlorine .
properties
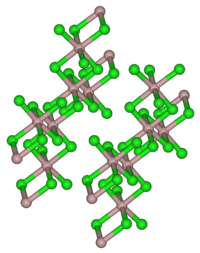
Thulium (III) chloride is a light yellow powder. Its hexahydrate is a light green hygroscopic solid. Both are soluble in water. Thulium (III) chloride has a monoclinic crystal structure with the space group C 2 / m (No. 12) corresponding to that of aluminum (III) chloride .
Individual evidence
- ↑ a b c d e Jean D'Ans, Ellen Lax: Paperback for chemists and physicists . 2007, ISBN 978-3-540-60035-0 , pp. 780 ( limited preview in Google Book search).
- ↑ a b c d e data sheet Thulium (III) chloride, anhydrous, powder, 99.9% trace metals basis from Sigma-Aldrich , accessed on April 30, 2012 ( PDF ).
- ↑ a b c d e Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 897.
- ↑ Web elements: Thulium
- ↑ Data sheet Thulium (III) chloride hexahydrate, 99.99% trace metals basis from Sigma-Aldrich , accessed on April 30, 2012 ( PDF ).



