Trichloroanilines
In chemistry, the trichloroanilines form a group of aromatic compounds that are derived from both aniline and chlorobenzene . The structure consists of a benzene ring with an attached amino group (–NH 2 ) and three chlorine (–Cl) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 6 H 4 Cl 3 N. All substances are sparingly soluble in water and toxic.
| Trichloroanilines | |||||||||||||
| Surname | 2,3,4-trichloroaniline | 2,3,5-trichloroaniline | 2,3,6-trichloroaniline | 2,4,5-trichloroaniline | 2,4,6-trichloroaniline | 3,4,5-trichloroaniline | |||||||
| other names | 1-amino-2,3,4-trichlorobenzene, 2,3,4-trichlorophenylamine |
1-amino-2,3,5-trichlorobenzene, 2,3,5-trichlorophenylamine |
1-amino-2,3,6-trichlorobenzene, 2,3,6-trichlorophenylamine |
1-amino-2,4,5-trichlorobenzene, 2,4,5-trichlorophenylamine |
1-amino-2,4,6-trichlorobenzene, 2,4,6-trichlorophenylamine |
1-amino-3,4,5-trichlorobenzene, 3,4,5-trichlorophenylamine |
|||||||
| Structural formula |

|

|

|
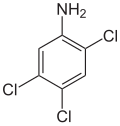
|

|

|
|||||||
| CAS number | 634-67-3 | 18487-39-3 | 88963-39-7 | 636-30-6 | 634-93-5 | 634-91-3 | |||||||
| PubChem | 12464 | 87676 | 184908 | 12487 | 12471 | 12469 | |||||||
| Molecular formula | C 6 H 4 Cl 3 N | ||||||||||||
| Molar mass | 196.46 g mol −1 | ||||||||||||
| Physical state | firmly | ||||||||||||
| Brief description | crystalline solids | ||||||||||||
| Melting point | 65-67 ° C | - | 93-94 ° C | 73-75 ° C | 97 ° C | ||||||||
| boiling point | 292 ° C | - | - | 270 ° C | 262 ° C | - | |||||||
| solubility | practically insoluble in water | ||||||||||||
|
GHS labeling |
|
|
|
||||||||||
| H and P phrases | 301-311-331-373-410 | see above | 301-311-331-373-410 | ||||||||||
| no EUH phrases | see above | no EUH phrases | |||||||||||
| no P-phrases | see above |
261-273-280 301 + 310-311-501 |
|||||||||||
use
Trichloroanilines are used as intermediate products in the production of other chemical compounds (e.g. 2,4,5-trichloroaniline for pigments and azo dyes ).
Tetra- and pentachloroanilines
Tetrachloroanilines (C 6 H 3 Cl 4 N) have four chlorine atoms on the benzene ring. They are produced by further chlorinating trichloroanilines (e.g. 2,3,4,6-tetrachloroaniline by chlorinating 2,4,5-trichloroaniline). There are three different isomers.
Pentachloroaniline is also known (C 6 H 2 Cl 5 N, M = 265.35 g mol −1 ).
Individual evidence
- ↑ a b c d Entry for CAS no. 634-67-3 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 18487-39-3 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c d Entry for CAS no. 636-30-6 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c d Entry for CAS no. 634-93-5 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 634-91-3 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ 2,4,5-Trichloroaniline BUA Summary (English). ( Memento of the original from May 21, 2003 in the Internet Archive ) Info: The archive link was automatically inserted and not yet checked. Please check the original and archive link according to the instructions and then remove this notice. (PDF; 591 kB).
- ↑ TU Darmstadt: Chemistry for Schoolchildren: Synthetic Organic Dyes (PDF; 625 kB) ( Memento from March 19, 2013 in the Internet Archive ), accessed on April 21, 2013.
- ↑ M. Mullin, G. Sawka, L. Safe, S. McCrindle, S. Safe: Synthesis of the Octa- and nonachlorobiphenyl isomers and congeners and their quantitation in commercial polychlorinated biphenyls and identification in human breast milk , in: J Anal Toxicol , 1981 , 5 (3) , pp. 138-142; PMID 6790872 .
- ↑ External identifiers or database links for pentachloroaniline : CAS number: 527-20-8, EC number: 208-410-3, ECHA InfoCard: 100.007.647 , PubChem : 10693 , ChemSpider : 10243 , Wikidata : Q27291304 .


