Uranium (IV) bromide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| __ U 4+ __ Br - | ||||||||||||||||
| Crystal system | ||||||||||||||||
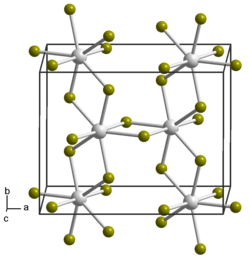
| Space group |
C 2 / m (No. 12) |
|||||||||||||||
| Lattice parameters |
a = 1092 pm |
|||||||||||||||
| General | ||||||||||||||||
| Surname | Uranium (IV) bromide | |||||||||||||||
| other names |
Uranium tetrabromide |
|||||||||||||||
| Ratio formula | UBr 4 | |||||||||||||||
| Brief description |
deep brown solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 557.65 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
5.55 g cm −3 |
|||||||||||||||
| Melting point |
519 ° C |
|||||||||||||||
| boiling point |
761 ° C |
|||||||||||||||
| solubility |
soluble in water and ethanol |
|||||||||||||||
| Hazard and safety information | ||||||||||||||||
 Radioactive |
||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Uranium (IV) bromide is a chemical compound made up of the elements uranium and bromine . It has the formula UBr 4 and belongs to the class of the bromides .
presentation
Uranium (IV) bromide can be obtained by reacting uranium with bromine at 600 ° C to 700 ° C.
Alternatively, the compound can be obtained from uranium (III) hydride by a two-step reaction . For this purpose, this is reacted with hydrogen bromide to form uranium (III) bromide and this with bromine at 300 ° C to form uranium (IV) bromide.
It is also possible to display it by reacting uranium (IV) oxide or uranium (V, VI) oxide with coal and bromine.
In addition, a few other display methods are known.
properties
Uranium (IV) bromide is a deep brown, very hygroscopic , crystalline solid. It has a monoclinic crystal structure with the space group C 2 / m (space group no. 12) and the lattice parameters a = 1092 pm, b = 869 pm, c = 705 pm and β = 93 ° 15 ′.
Individual evidence
- ↑ a b c d e f g h i j Georg Brauer (Ed.): Handbook of Preparative Inorganic Chemistry . 3., reworked. Edition. tape II . Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 1214 .
- ^ Lester R. Morss, Norman M. Edelstein, J. Fuger: The Chemistry of the Actinide and Transactinide Elements (Set Vol.1-6) . Springer Science & Business Media, 2010, ISBN 978-94-007-0211-0 , p. 1795 ( limited preview in Google Book search).
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 93rd Edition . CRC Press, 2012, ISBN 978-1-4398-8049-4 , pp. 97 ( limited preview in Google Book search).
- ↑ The hazards emanating from radioactivity do not belong to the properties to be classified according to the GHS labeling. With regard to other hazards, this substance has either not yet been classified or a reliable and citable source has not yet been found.
- ^ JJ Katz, Eugene Rabinowitch: The Chemistry of Uranium . 1961, ISBN 5-88135-966-6 , pp. 521 ( limited preview in Google Book search).
- ^ RM Douglass, Eugene Staritzky: Crystallographic Data. 153. Uranium Tetrabromide, UBr 4 In: Analytical Chemistry. 29, 1957, pp. 459-459, doi: 10.1021 / ac60123a607 .



