Zinc nitride
| Crystal structure | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||
| __ N 3− __ Zn 2+ | |||||||||||||
| General | |||||||||||||
| Surname | Zinc nitride | ||||||||||||
| other names |
Trizinc dinitride |
||||||||||||
| Ratio formula | Zn 3 N 2 | ||||||||||||
| Brief description |
black-gray solid |
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 224.2 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| density |
6.22 g cm −3 |
||||||||||||
| Melting point |
600 ° C (decomposition) |
||||||||||||
| solubility |
soluble in hydrochloric acid and sodium hydroxide with formation of ammonia |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Zinc nitride is an inorganic chemical compound of zinc from the group of nitrides . It was first synthesized in 1940 by R. Juza and H. Hahn.
Extraction and presentation
Zinc nitride can be obtained by reacting zinc or zinc oxide with ammonia .
It is also possible to manufacture by reacting zinc with nitrogen at 600 ° C.
properties
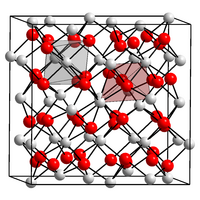
Zinc nitride is a blackish-gray solid that is largely stable in air. It has a cubic crystal structure. This is isostructural to that of magnesium nitride , calcium nitride and cadmium nitride, an anti- bixbyite structure with the space group Ia 3 (space group number 206) and the lattice parameter a = 9.7691 Å . The crystal structure is a derivative of that of calcium fluoride , with nitrogen and zinc atoms taking the position of calcium and three quarters of the fluorine positions. In this cubic structure, zinc atoms occupy the tetrahedral gaps of an approximately cubic packed arrangement of nitrogen atoms.
Individual evidence
- ↑ a b c d Georg Brauer (Ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 1031.
- ^ A b c Jean d'Ans, Ellen Lax, Roger Blachnik: Taschenbuch Fur Chemiker und Physiker . Springer DE, 1998, ISBN 3-642-58842-5 , pp. 810 ( limited preview in Google Book search).
- ↑ a b Zinc Nitride data sheet from AlfaAesar, accessed on May 8, 2013 ( PDF )(JavaScript required) .
- ↑ a b J. G. Zhao, LX Yang, SJ You, FY Li, CQ Jin, J. Liu: Structural stability of Zn 3 N 2 under high pressure . In: Physica B: Condensed Matter . tape 405 , no. 7 , April 2010, p. 1836–1838 , doi : 10.1016 / j.physb.2010.01.057 .
- ↑ a b R. K. Sharma: Chemistry Of Hydrides And Carbides . Discovery Publishing House, 2007, ISBN 81-8356-227-2 , pp. 313 ( limited preview in Google Book search).



