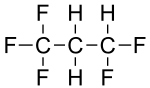
1,1,1,3,3-pentafluoropropane
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Surname | 1,1,1,3,3-pentafluoropropane | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 3 H 3 F 5 | |||||||||||||||||||||
| Brief description |
colorless gas with a faint odor |
|||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 134.05 g mol −1 | |||||||||||||||||||||
| Physical state |
gaseous |
|||||||||||||||||||||
| density |
1.32 g cm −3 |
|||||||||||||||||||||
| Melting point |
−103 ° C |
|||||||||||||||||||||
| boiling point |
15.3 ° C |
|||||||||||||||||||||
| Vapor pressure |
1227 hPa (20 ° C) |
|||||||||||||||||||||
| solubility |
|
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Global warming potential |
1032 (based on 100 years) |
|||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
1,1,1,3,3-Pentafluoropropane is a chemical compound from the group of aliphatic , saturated halogenated hydrocarbons .
Extraction and presentation
1,1,1,3,3-pentafluoropropane can be obtained by fluorinating 1,1,1,3,3-pentachloropropane (for example with antimony pentafluoride ).
properties
1,1,1,3,3-Pentafluoropropane is a colorless gas with a faint odor that is sparingly soluble in water. It has a high global warming potential with a value of 1032 based on a period of 100 years, which corresponds to 1032 times the global warming potential of carbon dioxide (CO 2 ).
use
1,1,1,3,3-pentafluoropropane is used as a foaming agent.
Individual evidence
- ↑ a b c d e f g h i j Entry on 1,1,1,3,3-pentafluoropropane in the GESTIS substance database of the IFA , accessed on November 9, 2018(JavaScript required) .
- ↑ a b c JACC 044: 1,1,1,3,3-pentafluoropropane (HFC-245fa) (CAS No. 460-73-1). European Center for Ecotoxicology, 2004, accessed November 10, 2018 .
- ↑ G. Myhre, D. Shindell et al .: Climate Change 2013: The Physical Science Basis . Working Group I contribution to the IPCC Fifth Assessment Report. Ed .: Intergovernmental Panel on Climate Change . 2013, Chapter 8: Anthropogenic and Natural Radiative Forcing, pp. 24-39; Table 8.SM.16 ( PDF ).
- ↑ Heng-Dao Quan, Hui-E Yang and a .: Preparation of 1,1,1,3,3-pentafluoropropane (HFC-245fa) by using a SbF5-attached catalyst. In: Journal of Fluorine Chemistry. 128, 2007, p. 190, doi : 10.1016 / j.jfluchem.2006.12.004 .
