2-bromonaphthalene
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
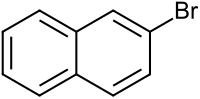
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 2-bromonaphthalene | |||||||||||||||
| other names |
β-naphthyl bromide |
|||||||||||||||
| Molecular formula | C 10 H 7 Br | |||||||||||||||
| Brief description |
yellow solid with a characteristic odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 207.07 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
52-55 ° C |
|||||||||||||||
| boiling point |
281-283 ° C |
|||||||||||||||
| solubility |
practically insoluble in water (8 mg l −1 at 25 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
2-Bromonaphthalene (β-naphthyl bromide) is a chemical compound that belongs to the substance class of substituted naphthalenes . It is isomeric to 1-bromonaphthalene .
Extraction and presentation
2-Bromonaphthalene can be prepared in a Sandmeyer reaction from 2-aminonaphthalene (β-naphthylamine) with copper (I) bromide as a reagent. The reaction takes place via the diazonium salt as an intermediate.
properties
2-Bromonaphthalene is a yellow solid.
use
2-Bromonaphthalene can be used for the synthesis of biaryls in a Suzuki cross-coupling reaction and for the preparation of a pentacarbonyl [methoxy (2-naphthyl) carbene] chromium complex.
Individual evidence
- ↑ a b c d e f g Entry on 2-bromonaphthalene in the GESTIS substance database of the IFA , accessed on February 24, 2017(JavaScript required) .
- ↑ H. Meislich, H. Nechamkin, J. Sharefkin: Organic Chemistry. (= Foam's outline ). McGraw Hill, Düsseldorf 1980, ISBN 978-0-070-63425-1 , p. 341.
- ↑ Data sheet 2-bromonaphthalene from Sigma-Aldrich , accessed on February 25, 2017 ( PDF ).
- ↑ Hans Joachim Stendel: Highly substituted hydrochinoid tricarbonyl (phenanthrene) chromium and tricarbonyl (triphenylene) chromium complexes: syntheses, structures and haptotropic metal migrations . Bonn 2005, DNB 974188557 , urn : nbn: de: hbz: 5N-04972 (dissertation, University of Bonn).

