3-penten-2-one
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
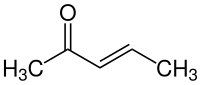
|
|||||||||||||
| Structural formula of the (E) isomer | |||||||||||||
| General | |||||||||||||
| Surname | 3-penten-2-one | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 5 H 8 O | ||||||||||||
| Brief description |
colorless liquid |
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 84.12 g mol −1 | ||||||||||||
| Physical state |
liquid |
||||||||||||
| density |
|
||||||||||||
| boiling point |
|
||||||||||||
| solubility |
soluble in water, acetone and ether ( E ) |
||||||||||||
| Refractive index |
1.4355 (20 ° C) |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| Toxicological data | |||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||
3-penten-2-one is a chemical compound from the group of ketones that occurs in two stereoisomeric forms.
Isomers
3-penten-2-one can have ( E ) - or ( Z ) -configuration at the double bond , consequently there are two isomers of the substance.
| Isomers of 3-penten-2-one | ||
| Surname | ( E ) -3-penten-2-one | ( Z ) -3-penten-2-one |
| other names | trans -3-penten-2-one | cis -3-penten-2-one |
| Structural formula |  |

|
| CAS number | 3102-33-8 | 3102-32-7 |
| 625-33-2 (unspec.) | ||
| EC number | - | - |
| 210-888-3 (unspec.) | ||
| ECHA info card | - | - |
| 100.009.899 (unspec.) | ||
| PubChem | 637920 | 5356572 |
| - (unspec.) | ||
| Wikidata | Q27161739 | Q27292477 |
| Q64774214 (unspec.) | ||
Occurrence
3-penten-2-one occurs naturally in the berries of two types of aronia melanocarpa . It has also been found in various other plants and foods such as tomatoes, cocoa, tea and potato chips.
Extraction and presentation
3-penten-2-one can be obtained by reacting 4-hydroxy-pentan-2-one with oxalic acid .
properties
3-penten-2-one is a colorless liquid with a fruity to pungent odor.
use
3-Penten-2-one can be used to synthesize other chemical compounds such as the alkaloids Senepodin G and Cermizin C. It is also used as a flavoring agent.
Individual evidence
- ↑ a b c d e f g Entry on 3-penten-2-one in the GESTIS substance database of the IFA , accessed on July 25, 2017(JavaScript required) .
- ^ A b Carl L. Yaws: The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals Physical Properties for More Than 54,000 Organic and Inorganic Chemical Compounds, Coverage for C1 to C100 Organics and Ac to Zr Inorganics . Gulf Professional Publishing, 2015, ISBN 978-0-12-801146-1 , pp. 72 ( limited preview in Google Book search).
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 97th Edition . CRC Press, 2016, ISBN 978-1-4987-5429-3 , pp. 440 ( limited preview in Google Book Search).
- ↑ a b c d data sheet 3-Penten-2-one, tech. 85% at AlfaAesar, accessed July 25, 2017 ( PDF )(JavaScript required) .
- ↑ a b Data sheet 3-Penten-2-one, 70% from Sigma-Aldrich , accessed on July 25, 2017 ( PDF ).
- ↑ a b c d George A. Burdock: Encyclopedia of Food and Color Additives . CRC Press, 1997, ISBN 978-0-8493-9414-0 , pp. 2997 ( limited preview in Google Book search).

