Aluminum triformate
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
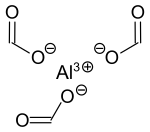
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Aluminum triformate | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 3 H 3 AlO 6 | ||||||||||||||||||
| Brief description |
white odorless solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 162.03 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.6475 g cm −3 |
||||||||||||||||||
| solubility |
easily soluble in water (200 g l −1 at ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Aluminum triformate is a chemical compound of aluminum from the group of carboxylic acid salts and one of the aluminum formates.
Extraction and presentation
Aluminum triformate can be obtained by reacting aluminum hydroxide with formic acid or by reacting barium formate with aluminum sulfate.
properties
Aluminum triformate is a white, odorless solid that is easily soluble in water. It decomposes when heated above 325 ° C.
use
Aluminum triformate is used as an aid in paper production, as a stain in the textile industry and as an astringent in cosmetics.
Individual evidence
- ↑ Entry on ALUMINUM TRIFORMATE in the CosIng database of the EU Commission, accessed on December 28, 2019.
- ↑ a b c d e f g Entry on aluminum triformate in the GESTIS substance database of the IFA , accessed on December 9, 2019(JavaScript required) .
- ↑ Moxi Xue, Baoyu Gao et al. a .: Aluminum formats (AF): Synthesis, characterization and application in dye wastewater treatment. In: Journal of Environmental Sciences. 74, 2018, p. 95, doi : 10.1016 / j.jes.2018.02.013 .
- ↑ Google Patents: US5468892A - Process for the production of an aluminum triformate solution containing alkali metal and / or alkaline earth metal, highly concentrated aluminum triformate solutions containing alkali and / or alkaline earth metal, and their use - Google Patents , accessed on 9. December 2019
- ↑ Werner Baumann, Herberg-Liedtke: Paper chemicals data and facts on environmental protection . Springer-Verlag, 2013, ISBN 978-3-642-58040-6 , pp. 371 ( limited preview in Google Book search).
