Ammonium hydrogen difluoride
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
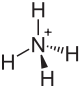 |
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Ammonium hydrogen difluoride | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | NH 4 HF 2 | ||||||||||||||||||
| Brief description |
colorless solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 57.04 g · mol -1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.5 g cm −3 |
||||||||||||||||||
| Melting point |
124.6 ° C |
||||||||||||||||||
| boiling point |
230 ° C (decomposition) |
||||||||||||||||||
| solubility |
good in water (630 g l −1 at 20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| MAK |
1 mg m −3 |
||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Ammonium hydrogen difluoride is an acidic ammonium salt of hydrofluoric acid .
properties
Ammonium hydrogen difluoride forms colorless, easily deliquescent crystals that are easily soluble in water. It etches glass and is also used in industry for stain removal and in the petroleum industry to work through granite layers. The species attacking the silicon dioxide is the HF 2 - anion. It is the active component of various etching mixtures for the selective etching of silicon dioxide layers in the semiconductor industry .
Reactions
When heated strongly, it breaks down into ammonia and hydrogen fluoride.
synthesis
Ammonium hydrogen difluoride is prepared by reacting aqueous ammonia solution with hydrofluoric acid in a molar ratio of 1: 2 and then crystallizing from water.
Precautions
Ammonium hydrogen difluoride is corrosive and toxic. Avoid contact with skin and eyes. Do not swallow.
disposal
Precipitate dissolved fluoride as calcium fluoride .
See also
- Three-center bond , to the hydrogen difluoride anion
Individual evidence
- ↑ a b c d e f g h i j Entry on ammonium hydrogen difluoride in the GESTIS substance database of the IFA , accessed on January 10, 2017(JavaScript required) .
- ↑ Entry on ammonium hydrogendifluoride in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on February 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ^ The Electrochemical Society: Cleaning Technology in Semiconductor Device Manufacturing. The Electrochemical Society, 2000, ISBN 978-1-566-77259-4 , p. 248 ( limited preview in Google book search).
- ^ Tadahiro Ohmi: Scientific Wet Process Technology for Innovative LSI / FPD Manufacturing. CRC Press, 2016, ISBN 978-1-420-02686-3 , p. 178 ( limited preview in Google book search).
- ↑ Alain Tressaud: Functionalized Inorganic Fluorides Synthesis, Characterization and Properties of Nanostructured Solids . John Wiley & Sons, 2010, ISBN 0-470-66075-9 , pp. 210 ( limited preview in Google Book search).



