Fumarylacetoacetase
| Fumarylacetoacetate hydrolase | ||
|---|---|---|

|
||
| Ribbon model of the mouse FAA with Ca 2+ (yellow), Ni 2+ (orange), products (purple) according to PDB 1QCO | ||
| Properties of human protein | ||
| Mass / length primary structure | 46.4 kDa / 419 amino acids | |
| Secondary to quaternary structure | Homodimer | |
| Cofactor | Ca 2+ , Mg 2+ | |
| Identifier | ||
| Gene names | FAH ; FAA | |
| External IDs | ||
| Enzyme classification | ||
| EC, category | 3.7.1.2 , hydrolase | |
| Response type | hydrolysis | |
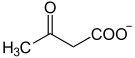
| Substrate | 4-fumarylacetoacetate + H 2 O | |
| Products | Fumarate + acetoacetate | |
| Occurrence | ||
| Homology family | Fumarylacetoacetate hydrolase | |
| Parent taxon | Creature | |
| Orthologue | ||
| human | House mouse | |
| Entrez | 2184 | 14085 |
| Ensemble | ENSG00000103876 | ENSMUSG00000030630 |
| UniProt | P16930 | P35505 |
| Refseq (mRNA) | NM_000137 | NM_010176 |
| Refseq (protein) | NP_000128 | NP_034306 |
| Gene locus | Chr 15: 80.15 - 80.19 Mb | Chr 7: 84.59 - 84.61 Mb |
| PubMed search | 2184 |
14085
|
Fumarylacetoacetase (FAA, FAH) is the name of the enzyme that hydrolyzes 4-fumarylacetoacetate into fumarate and acetoacetate . This reaction is the last step in the breakdown of the amino acid tyrosine and is carried out in all living things. A lack or absence of the enzyme in humans results in a metabolic disease , the Type I tyrosinemia . The cause is always a mutation in the FAH gene that codes for the enzyme .
biosynthesis
FAA is mainly produced in the liver and kidneys , but also to a small extent in any other tissue . The FAH gene is located on chromosome 15 and comprises 33,340 base pairs and 14 exons . After transcription , mRNA is produced with a length of 1,447 bases, the translation of which results in a protein with 419 amino acids .
Catalyzed reaction
Fumarylacetoacetate is hydrolyzed to fumarate and acetoacetate, the enzyme also needs calcium and magnesium ions as cofactors . FAA is also able to hydrolyze other 3,5- and 2,4-dioxocarboxylic acids.