Hafnium (IV) bromide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
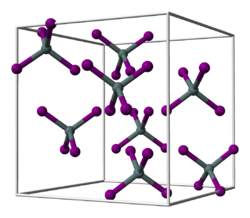
|
||||||||||||||||
| __ Hf 4+ __ Br - | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Hafnium (IV) bromide | |||||||||||||||
| other names |
Hafnium tetrabromide |
|||||||||||||||
| Ratio formula | HfBr 4 | |||||||||||||||
| Brief description |
white solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 498.11 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
4.9 g cm −3 (25 ° C) |
|||||||||||||||
| Melting point |
424 ° C (under pressure) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Hafnium (IV) bromide is an inorganic chemical compound of hafnium from the group of bromides .
Extraction and presentation
Hafnium (IV) bromide can be obtained by bromination of hafnium with bromine at 320-350 ° C.
It is also possible to produce it by reacting hafnium (IV) oxide with carbon and bromine.
properties
Hafnium (IV) bromide is a white solid. When exposed to water, it hydrolyzes completely. Its crystal structure corresponds to that of tin (IV) iodide with the space group Pa 3 (space group no. 205) . It sublimes at 317 ° C.
Individual evidence
- ↑ a b Roger Blachnik (Ed.): Pocket book for chemists and physicists . Volume III: Elements, Inorganic Compounds and Materials, Minerals . founded by Jean d'Ans, Ellen Lax. 4th, revised and revised edition. Springer, Berlin 1998, ISBN 3-540-60035-3 , pp. 478 f . ( limited preview in Google Book search).
- ↑ a b c d e data sheet hafnium (IV) bromide, anhydrous, powder, 99.99% (purity excludes ~ 0.2% zirconium.) At Sigma-Aldrich , retrieved on May 4, 2013 ( PDF ).
- ↑ a b Georg Brauer (Ed.), With the collaboration of Marianne Baudler u a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 1358.
- ↑ Georg Brauer (Ed.), With the collaboration of Marianne Baudler a. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 1359.


