Caryophyllene
| Caryophyllene | ||||
| Surname | α-caryophyllene | β-caryophyllene | ||
| Structural formula |
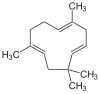
|

|
||
| other names | Humulene | E-BCP | ||
| CAS number | 6753-98-6 | 87-44-5 | ||
| PubChem | 5281520 | 5281515 | ||
| Molecular formula | C 15 H 24 | |||
| Molar mass | 204.36 g mol −1 | |||
| Physical state | liquid | |||
| Brief description | yellowish-green liquid | Liquid with an odor similar to clove to turpentine |
||
| boiling point | 166-168 ° C | 260-261 ° C | ||
| density | 0.90-0.91 g cm -3 | 0.89 g cm −3 | ||
|
GHS labeling |
|
|
||
| H and P phrases | 315-319-335 | no H-phrases | ||
| no EUH phrases | no EUH phrases | |||
| 261-305 + 351 + 338 | no P-phrases | |||
Caryophyllene (stress on the penultimate syllable: Caryophyll e ne ) are sesquiterpenes that are contained in essential oils . There are α-caryophyllene (humulene) and β-caryophyllene (abbreviated E-BCP for ( E ) - B eta C aryo p hyllen).
Occurrence
α-Caryophyllene has a monocyclic structure and occurs in cloves and many types of basil . β-Caryophyllene has a bicyclic structure; it is found in basil , cannabis , rosemary , cinnamon , oregano , caraway , hops and pepper .
effect
Sergio Rasmann from the University of Neuchâtel discovered in 2005 that E-BCP is secreted by maize plants when predators (e.g. corn rootworm ) eat their roots. This attracts predatory nematodes ( Heterorhabditis megidis ), which attack the predators .
β-caryophyllene oxidizes in the air to caryophyllene oxide , a substance with a weak allergenic effect; after five weeks about half of the caryophylline is converted.
pharmacology
β-caryophyllene is a CB 2 - cannabinoid and reduces inflammation. Since this active ingredient, unlike other known substances that act on the same CB 2 receptor but not on the CB 1 receptor , does not have an intoxicating effect, this natural substance can be used as a starting point for the development of new drugs.
Individual evidence
- ↑ a b c Data sheet α-Caryophyllene at Good Scents Company , accessed June 7, 2017.
- ↑ Entry on Caryophyllene. In: Römpp Online . Georg Thieme Verlag, accessed on December 25, 2014.
- ↑ a b Datasheet beta-Caryophyllene from Good Scents Company , accessed June 7, 2017.
- ↑ Data sheet a-Humulene from Sigma-Aldrich , accessed on April 27, 2011 ( PDF ).
- ↑ Data sheet (-) - trans-Caryophyllene from Sigma-Aldrich , accessed on April 27, 2011 ( PDF ).
- ↑ Jürg Gertsch, Marco Leonti, Stefan Raduner, Ildiko Racz, Jian-Zhong Chen: Beta-caryophyllene is a dietary cannabinoid . In: Proceedings of the National Academy of Sciences . tape 105 , no. July 26 , 2008, p. 9099-9104 , doi : 10.1073 / pnas.0803601105 , PMID 18574142 , PMC 2449371 (free full text).
- ↑ Spectrum of Science: Parasite Attractant Against Corn Pests . November 2009, pp. 20-22.
- ↑ M. Skold et al. (2006): The fragrance chemical beta-caryophyllene-air oxidation and skin sensitization. In: Food Chem. Toxicol. Vol. 44, No. 4, pp. 538-545; PMID 16226832 .
- ↑ Gertsch, J. et al. (2008): Beta-caryophyllene is a dietary cannabinoid. In: Proc. Natl. Acad. Sci. USA 105 (26): pp. 9099-9104; PMID 18574142 ; doi : 10.1073 / pnas.0803601105 .
- ↑ Deutsches Ärzteblatt : Oregano pizza spice helps against inflammation , June 24, 2008.

