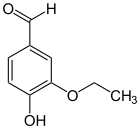
Isoethylvanillin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Isoethylvanillin | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 9 H 10 O 3 | ||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 166.17 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
127-128 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Isoethylvanillin ( 4-ethoxy-3-hydroxybenzaldehyde ) is an organic chemical compound with the empirical formula C 9 H 10 O 3 . It is a derivative of benzaldehyde with an additional hydroxy and an ethoxy group . It is an isomer to ethyl vanillin , from which it differs only in the position of the ethoxy group. Instead of position 3, this is found here in position 4. Hydroxy and ethoxy groups swap places compared to ethyl vanillin. But it is also derived from isovanillin by exchanging the methyl group for an ethyl group.
Isoethylvanillin is formed when 3,4-dihydroxybenzaldehyde is heated with diethyl sulfate in ethanolic KOH .