Lavandulol
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
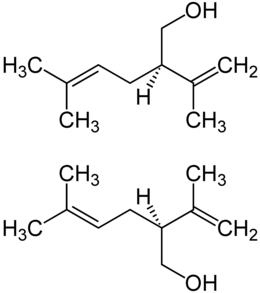
|
|||||||||||||||||||
| ( R ) -Lavandulol (top) and ( S ) -Lavandulol (bottom) | |||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Lavandulol | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 10 H 18 O | ||||||||||||||||||
| Brief description |
colorless liquid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 154.25 g · mol -1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
0.878 g cm −3 |
||||||||||||||||||
| Refractive index |
1.47 (20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||||||||
Lavandulol is the common name of the acyclic compound (±) -2-isopropenyl-5-methyl-4-hexen-1-ol.
Occurrence
Lavandulol is a terpene found in lavender oil .
properties
Lavandulol is chiral . Thus there are the stereoisomers ( R ) -Lavandulol and ( S ) -Lavandulol. ( R ) -Lavandulol has a faint floral herbal smell with a lime-like, fresh lemon-fruit nuance. ( S ) -Lavandulol, on the other hand, has a very weak taste. Both are clear liquids.
use
( R ) - and ( S ) -Lavandulol and their esters are important components in the perfume industry and have recently gained significance in pheromone research . Lavandulol has also been identified as a reproductive pheromone in mealybugs .
Individual evidence
- ↑ a b Hiroyuki Sakauchi, Hiromasa Kiyota, Shin-ya Takigawa, Takayuki Oritani, Shigefumi Kuwahara: Enzymatic Resolution and Odor Description of Both Enantiomers of Lavandulol, a Fragrance of Lavender Oil . Chemistry & Biodiversity, 2 (9), 1183–1186, 2005. doi : 10.1002 / cbdv.200590088 (quoted from leffingwell.com )
- ↑ a b c Data sheet (±) -Lavandulol from Sigma-Aldrich , accessed on May 29, 2011 ( PDF ).
