Lutidines
In chemistry, the lutidines or dimethylpyridines form a group of organic compounds that belong to the heterocycles (more precisely: heteroaromatics ). They consist of a pyridine ring that is substituted with two methyl groups. Their different arrangement results in six constitutional isomers with the empirical formula C 7 H 9 N. 2,6-lutidine is the best-known isomer .
properties
The lutidines are colorless to yellow liquids with a characteristic odor and a slightly oily consistency. They are soluble in water, ethanol , diethyl ether and acetone . The compounds are classified as harmful , two of the isomers are classified as toxic .
Oxidation of the methyl groups produces the pyridinedicarboxylic acids . The name is only adopted on the product for the 2,4-isomer: The pyridine-2,4-dicarboxylic acid is called lutidic acid , the names of the other five isomers have a different origin.
| Lutidines | |||||||||||||||||||
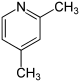
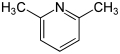
| Surname | 2,3-lutidine | 2,4-lutidine | 2,5-lutidine | 2,6-lutidine | 3,4-lutidine | 3,5-lutidine | |||||||||||||
| other names | 2,3-dimethylpyridine | 2,4-dimethylpyridine | 2,5-dimethylpyridine | 2,6-dimethylpyridine | 3,4-dimethylpyridine | 3,5-dimethylpyridine | |||||||||||||
| Structural formula |

|
|

|
|

|

|
|||||||||||||
| CAS number | 583-61-9 | 108-47-4 | 589-93-5 | 108-48-5 | 583-58-4 | 591-22-0 | |||||||||||||
| PubChem | 11420 | 7936 | 11526 | 7937 | 11417 | 11565 | |||||||||||||
| Molecular formula | C 7 H 9 N | ||||||||||||||||||
| Molar mass | 107.16 g mol −1 | ||||||||||||||||||
| Physical state | liquid | ||||||||||||||||||
| Brief description | colorless to yellow liquids with a characteristic odor and a slightly oily consistency | ||||||||||||||||||
| Melting point | −15 ° C | −60 ° C | −15 ° C | −6 ° C | −6 ° C | −9 ° C | |||||||||||||
| boiling point | 162-163 ° C | 159 ° C | 157 ° C | 144 ° C | 163-164 ° C | 169-170 ° C | |||||||||||||
|
pK s value (of the conjugate acid BH + ) |
6.57 | 6.77 | 6.40 | 6.60 | 6.46 | ||||||||||||||
| solubility | 95 g l −1 | 350 g l −1 | soluble | 33 g l −1 | 33 g l −1 | ||||||||||||||
|
GHS labeling |
|
|
|
|
|
|
|||||||||||||
| H and P phrases |
226-302-312-331 315-319-335 |
226-301 |
226-302-312 315-319-332-335 |
226-302-315-319-335 | 226-302-310 |
226-302-312-332 315-318-335 |
|||||||||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | ||||||||||||||
|
261-280-304 + 340 305 + 351 + 338-310 |
210-262-280-309 + 310 | 261-280-305 + 351 + 338 | 210-260-302 + 352-305 + 351 + 338 | 210-280-302 + 352-309 + 310 | 261-280-305 + 351 + 338 | ||||||||||||||
See also
Individual evidence
- ↑ Entry on Lutidine. In: Römpp Online . Georg Thieme Verlag, accessed on November 25, 2014.
- ↑ a b c d Entry for CAS no. 583-61-9 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c d Entry for CAS no. 108-47-4 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 589-93-5 in the GESTIS substance database of the IFA , accessed on July 15, 2013(JavaScript required) .
- ↑ a b c d Entry for CAS no. 108-48-5 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c d Entry for CAS no. 583-58-4 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c d Entry for CAS no. 591-22-0 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .



