Methyl styrenes
The methyl styrenes (also called vinyl toluenes or methyl vinyl benzenes) form a group of substances in chemistry that is derived from both styrene and toluene . The structure consists of a benzene ring with attached ethene (-CH = CH 2 ) and methyl groups (-CH 3 ) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 9 H 10 . They are mainly to be regarded as methyl-substituted styrenes. In addition, there are two isomeric compounds, isopropenylbenzene (α-methylstyrene) and 1-propenylbenzene (β-methylstyrene), in which the methyl group is attached to the ethene group and not to the benzene ring.
properties
Methyl styrenes are colorless liquids with an unpleasant odor.
| Methyl styrene | |||||||||||||||||||
| Surname | 2-methylstyrene | 3-methylstyrene | 4-methylstyrene | α-methylstyrene | β-methylstyrene | ||||||||||||||
| other names |
o -Methylstyrene, 2-vinyltoluene, 1-methyl-2-vinylbenzene |
m -methylstyrene, 3-vinyltoluene |
p -methylstyrene, 4-vinyltoluene |
2-phenylpropene isopropenylbenzene 2-phenyl-1-propene β-phenylpropylene |
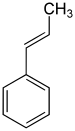
1-propenylbenzene | ||||||||||||||
| Structural formula |  |
 |
 |
 |
|
||||||||||||||
| CAS number | 611-15-4 | 100-80-1 | 622-97-9 | 98-83-9 | 637-50-3 ( cis -form) 873-66-5 ( trans -form) |
||||||||||||||
| 25013-15-4 (mixture of isomers) | |||||||||||||||||||
| PubChem | 11904 | 7529 | 12161 | 7407 |
252325 252324 |
||||||||||||||
| Molecular formula | C 9 H 10 | ||||||||||||||||||
| Molar mass | 118.18 g mol −1 | ||||||||||||||||||
| Physical state | liquid | ||||||||||||||||||
| Melting point | −69 ° C | −82 ° C | −34 ° C | −23.2 ° C | −27 ° C | ||||||||||||||
| boiling point | 171 ° C | 170 ° C | 170-175 ° C | 166 ° C | 172-173 ° C 175 ° C |
||||||||||||||
| density | 0.91 g cm −3 | 0.90 g cm −3 | 0.90 g cm −3 | 0.91 g cm −3 | 0.91 g cm −3 | ||||||||||||||
| Vapor pressure | 2.66 mbar (20 ° C) | 3.5 mbar (20 ° C) | 1.7 mbar (20 ° C) | 2.9 mbar (20 ° C) | |||||||||||||||
| solubility | practically insoluble in water | ||||||||||||||||||
|
GHS labeling |
|
|
|
|
|
||||||||||||||
| H and P phrases | 226-332-411 | 226-304-315-319-332-335 | 226-304-315-319-335 | 226-319-335-411 | 226-315-319-335 | ||||||||||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | |||||||||||||||
| 210-240-273-280 | 261-301 + 310-305 + 351 + 338-331 | 261-301 + 310-305 + 351 + 338-331 | 261-273-305 + 351 + 338 |
210-261-280-302 + 352 305 + 351 + 338 |
|||||||||||||||
| MAK | Switzerland: 35 ml m −3 or 172 mg m −3 | ||||||||||||||||||
use
2-methylstyrene is used as a modifier of alkyd resins ; for example, it is added to polystyrene polymerization in order to increase the heat resistance of polystyrene . Alpha-methylstyrene is used to manufacture plasticizers for plastics, paints, waxes, etc. as well as modified polyester and alkyd resins. Poly- alpha- methylstyrene (CAS number: 25014-31-7) can be obtained through polymerization .
safety instructions
If unstabilized, there is a risk of polymerization . The monomers are therefore stabilized and usually marketed as a mixture of isomers. When heated, the stabilizer (e.g. 3,5-di- tert- butyl-pyrocatechol ) can become ineffective and vapors of methyl styrenes can form, which can form an explosive mixture with air.
Individual evidence
- ↑ Entry on methyl styrene, isomers in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c d e f Entry on 2-methylstyrene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c d e f Entry on 3-methylstyrene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c d e f Entry on 4-methylstyrene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c d e f Entry on alpha-methylstyrene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c Entry on beta-methylstyrene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ a b c Entry on trans-propenylbenzene in the GESTIS substance database of the IFA , accessed on May 14, 2017(JavaScript required) .
- ↑ Swiss Accident Insurance Fund (Suva): Limit values - current MAK and BAT values (search for methylstyrene (all isomers) ), accessed on March 4, 2020.



