N- phenylhydroxylamine
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
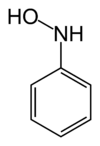
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | N- phenylhydroxylamine | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 6 H 7 NO | |||||||||||||||
| Brief description |
yellow solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 109.12 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.214 g cm −3 |
|||||||||||||||
| Melting point |
80-84 ° C |
|||||||||||||||
| boiling point |
215.8 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
N- phenylhydroxylamine is a chemical compound from the group of hydroxylamine derivatives .
Extraction and presentation
N- phenylhydroxylamine can be produced by reducing nitrobenzene with zinc in the presence of ammonium chloride.
In the human body, the compound occurs as a metabolite after exposure to nitrobenzene when it is broken down by intestinal bacteria .
properties
N- phenylhydroxylamine is a yellow, flammable solid.
Individual evidence
- ↑ a b c d e f g h i Entry for CAS no. 100-65-2 in the GESTIS substance database of the IFA , accessed on January 1, 2014(JavaScript required) .
- ↑ a b Eug. Bamberger: About the phenylhydroxylamine. In: Reports of the German Chemical Society. 27, 1894, pp. 1548-1557, doi : 10.1002 / cber.18940270276 .
- ^ Rainer Koch: Environmental chemicals: Physico-chemical data, toxicities, boundary and ... John Wiley & Sons, 2008, ISBN 3-527-62481-3 , p. 295 ( limited preview in Google Book search).

