Platinum (II) iodide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
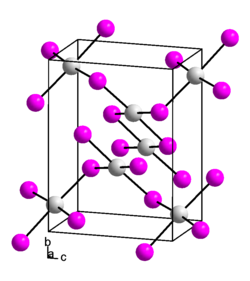
|
||||||||||||||||
| __ Pt 2+ __ I - | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Platinum (II) iodide | |||||||||||||||
| other names |
Platinum diiodide |
|||||||||||||||
| Ratio formula | PtI 2 | |||||||||||||||
| Brief description |
black solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 448.90 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
325 ° C (decomposition) |
|||||||||||||||
| solubility |
almost insoluble in water and alkali iodides |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Platinum (II) iodide is an inorganic chemical compound of platinum from the group of iodides .
Extraction and presentation
Platinum (II) iodide can be obtained by reacting platinum with iodine at 400 ° C (formation of the cubic form) or by heating potassium hexaiodoplatinate with water at 240 ° C (formation of the monoclinic form).
properties
Platinum (II) iodide is a black solid that is insoluble in water, alkali iodides, nitric acid , hydrochloric acid or sulfuric acid . It comes in two modifications, one with a cubic and one with a monoclinic crystal structure .
Individual evidence
- ↑ a b c Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1716.
- ↑ a b c d e f data sheet Platinum (II) iodide, Premion®, 99.99% (metals basis), Pt 43.0% min from AlfaAesar, accessed on August 30, 2013 ( PDF )(JavaScript required) .
