Rubidium propionate
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
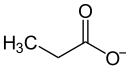 |
||||||||||
| General | ||||||||||
| Surname | Rubidium propionate | |||||||||
| other names |
Rubidium Propanoate (according to IUPAC ) |
|||||||||
| Molecular formula | C 3 H 5 O 2 Rb | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 158.54 g · mol -1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
350 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Rubidium propionate is the rubidium salt of propionic acid .
Manufacturing
Rubidium propionate can be produced from rubidium hydroxide and propionic acid by the salt formation reaction .
The synthesis from rubidium carbonate and propionic acid is also possible with the evolution of carbon dioxide .
properties
Rubidium propionate can occur in three different crystal structures . The phase transitions take place at 564.3 K and 317 K.
Individual evidence
- ↑ a b Entry on rubidium propionate . In: P. J. Linstrom, W. G. Mallard (Eds.): NIST Chemistry WebBook, NIST Standard Reference Database Number 69 . National Institute of Standards and Technology , Gaithersburg MD .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ P. Ferloni, M. Sanesi, P. Franzosini: Phase transitions in the alkali C 1 -n C 4 alkanoates. In: Journal of Nature Research A . 30, 1975, pp. 1447-1454 ( PDF , free full text).

