Thallium (I) oxide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| __ Tl + __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Thallium (I) oxide | |||||||||||||||
| other names |
Dithallium monoxide |
|||||||||||||||
| Ratio formula | Tl 2 O | |||||||||||||||
| Brief description |
black solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 424.77 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
9.52 g cm −3 |
|||||||||||||||
| Melting point |
579 ° C |
|||||||||||||||
| boiling point |
1080 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Thallium (I) oxide is an inorganic chemical compound of thallium from the group of oxides .
Extraction and presentation
Thallium (I) oxide can be prepared by heating thallium (I) hydroxide to over 100 ° C or thallium (III) oxide to over 700 ° C in the absence of air.
properties
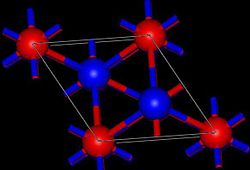
Thallium (I) oxide is a very hygroscopic black solid that is easily soluble in water with the formation of thallium (I) hydroxide. It has a crystal structure that is to be understood as a triple polytype form of the anti- cadmium (II) iodide type. It is a trigonal crystal structure with the space group R 3 m (space group no. 166) .
Individual evidence
- ↑ a b c d e f Jean D'Ans, Ellen Lax: Pocket book for chemists and physicists . Springer DE, 1997, ISBN 3-540-60035-3 , pp. 776 ( limited preview in Google Book search).
- ↑ David R. Lide: CRC Handbook of Chemistry and Physics . CRC Press, 2012, ISBN 1-4398-8049-2 , pp. 4–94 ( limited preview in Google Book Search).
- ↑ Entry on thallium compounds, inorganic, soluble in the GESTIS substance database of the IFA , accessed on November 26, 2013(JavaScript required) .
- ↑ Simon Aldridge, Anthony J. Downs: The Group 13 Metals Aluminum, Gallium, Indium and Thallium: Chemical ... John Wiley & Sons, 2011, ISBN 0-470-97668-3 , pp. 325 ( limited preview in Google Book Search).
- ↑ H. Sabrowsky: To represent and crystal structure of Tl 2 O . In: Journal for inorganic and general chemistry , 381 (3), 1971, pp. 266-279. doi : 10.1002 / zaac.19713810305



