Indium (III) oxide
| Crystal structure | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
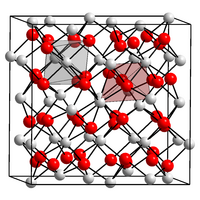
|
|||||||||||||||||||
| __ In 3+ __ O 2− | |||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Indium (III) oxide | ||||||||||||||||||
| other names |
Diindium trioxide |
||||||||||||||||||
| Ratio formula | In 2 O 3 | ||||||||||||||||||
| Brief description |
yellow powder |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 277.64 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
7.18 g cm −3 |
||||||||||||||||||
| Melting point |
1910 ° C |
||||||||||||||||||
| solubility |
almost insoluble in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Indium (III) oxide is a chemical compound of indium and oxygen . The air-stable, light yellow oxide is a semiconductor .
Extraction and presentation
Indium (III) oxide is formed from the elements at high temperatures under a blue flame .
Indium (III) oxide is also formed from indium (III) hydroxide when heated.
properties
There are two modifications of the light yellow colored indium (III) oxide. The α-modification, which is stable under normal conditions, changes under pressure at 1000 ° C into a β-modification. α-Indium (III) oxide crystallizes in a structure which can be derived from the calcium fluoride structure and in which there are fluoride vacancies. β-indium (III) oxide has a corundum structure .
Thin-film layers of indium (III) oxide, which are doped with chromium (In 2-x Cr x O 3 ), have ferromagnetic properties and are among the magnetic semiconductors .
use
Indium (III) oxide is mainly used to make indium tin oxide . This is an important part of liquid crystal displays and touch screens . The mixed oxide of manganese , yttrium and indium results in a brilliant blue pigment, the YInMn blue . The general formula is YIn 1-x Mn x O 3 If it does not contain manganese, the oxide is colorless, if it does not contain indium, it is black.
Individual evidence
- ↑ a b c d e data sheet Indium (III) oxide, 99.994% (metals basis) from AlfaAesar, accessed on December 7, 2019 ( PDF )(JavaScript required) .
- ^ O. Knacke, O. Kubaschewski, K. Hesselmann (eds.): Thermochemical properties of inorganic substances I. 2nd edition, Springer, Berlin, 1991.
- ↑ Nicole Hemsoth: MIT Material Puts New Spin on Electronics. In: hpcwire.com. May 6, 2006, accessed October 29, 2008 .
- ↑ Andrew E. Smith, Hiroshi Mizoguchi, Kris Delaney, Nicola A. Spaldin, Arthur W. Sleight, MA Subramanian: Mn in Trigonal Bipyramidal Coordination: A New Blue Chromophore. In: Journal of the American Chemical Society . 131, 2009, pp. 17084-17086, doi: 10.1021 / ja9080666 .
literature
- AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 .

