UDP glucuronate decarboxylase
| UDP glucuronate decarboxylase | ||
|---|---|---|
| Properties of human protein | ||
| Mass / length primary structure | 420 amino acids | |
| Cofactor | NAD | |
| Isoforms | 3 | |
| Identifier | ||
| Gene names | UXS1 SDR6E1; UGD | |
| External IDs | ||
| Enzyme classification | ||
| EC, category | 4.1.1.35 , lyase | |
| Response type | Splitting off of CO 2 | |
| Substrate | UDP glucuronate | |
| Products | UDP-xylose + CO 2 | |
| Occurrence | ||
| Parent taxon | Eukaryotes | |
| Orthologue | ||
| human | House mouse | |
| Entrez | 80146 | 67883 |
| Ensemble | ENSG00000115652 | ENSMUSG00000057363 |
| UniProt | Q8NBZ7 | Q91XL3 |
| Refseq (mRNA) | NM_001253875 | NM_026430 |
| Refseq (protein) | NP_001240804 | NP_080706 |
| Gene locus | Chr 2: 106.09 - 106.19 Mb | Chr 1: 43.75 - 43.83 Mb |
| PubMed search | 80146 |
67883
|
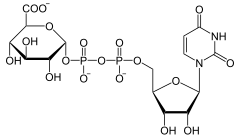
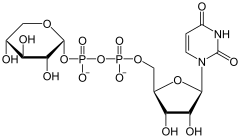
The UDP-glucuronate decarboxylase (UGD) is the enzyme that in eukaryotes the elimination of carbon dioxide from UDP-glucuronate catalyzed . The resulting UDP-xylose is necessary in plants for the biosynthesis of xylose and other pentoses . Vertebrates only need UDP-xylose for the production of glycosaminoglycans ( cartilage ). UGD is localized in the membrane of the Golgi apparatus .
Catalyzed reaction
UDP-α- D -glucuronate is converted to UDP-α- D -xylose.
Individual evidence
- ↑ Homologues at OMA
- ↑ UniProt Q8NB27
- ^ Moriarity JL, Hurt KJ, Resnick AC, et al. : UDP-glucuronate decarboxylase, a key enzyme in proteoglycan synthesis: cloning, characterization, and localization . In: J. Biol. Chem. . 277, No. 19, May 2002, pp. 16968-75. doi : 10.1074 / jbc.M109316200 . PMID 11877387 .
Web links
Wikibooks: Biochemistry and Pathobiochemistry: Uronic Acid Metabolism - Learning and Teaching Materials