Tungsten (VI) oxide tetrabromide
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
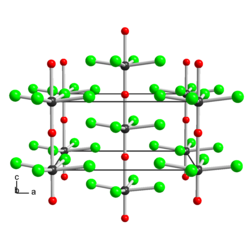
|
||||||||||
| __ W 6+ __ Br - __ O 2− | ||||||||||
| General | ||||||||||
| Surname | Tungsten (VI) oxide tetrabromide | |||||||||
| Ratio formula | WOBr 4 | |||||||||
| Brief description |
brown-black solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 519.46 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| density |
5.5 g cm −3 |
|||||||||
| Melting point |
277 ° C |
|||||||||
| boiling point |
327 ° C |
|||||||||
| solubility |
reacts with water |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Tungsten (VI) oxide tetrabromide is an inorganic chemical compound of tungsten from the group of oxide bromides .
Extraction and presentation
Tungsten (VI) oxide tetrabromide can be obtained by reacting tungsten (VI) oxide with tungsten and bromine or tungsten (VI) bromide .
It is also possible to obtain tungsten (VI) oxide by heating with tetrabromomethane at 440 ° C or with boron (III) bromide at 130 ° C, as well as reacting tungsten (III) bromide with liquid sulfur dioxide .
It is also formed during the decomposition of tungsten (VI) dioxide dibromide WO 2 Br 2 at 200 ° C.
properties
Tungsten (VI) oxide tetrabromide is a black-brown, crystalline, very moisture-sensitive solid that instantly decomposes in water. It is soluble in concentrated hydrochloric acid , 1,4-dioxane and acetone . It has a tetragonal crystal structure isotype with tungsten (VI) oxide tetrachloride (a = 896 pm, c = 393 pm).
Individual evidence
- ↑ a b c d e f Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1571.
- ↑ a b c W. M. Haynes (Ed.): CRC handbook of chemistry and physics. A ready-reference book of chemical and physical data . founded by David R. Lide. 93rd edition. CRC Press, Boca Raton 2012, ISBN 978-1-4398-8049-4 , pp. 4–97 (English, limited preview in Google Book Search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ Arnold F. Holleman, Egon Wiberg, Nils Wiberg: Inorganic Chemistry . Academic Press, 2001, ISBN 0-12-352651-5 , pp. 1387 ( limited preview in Google Book Search).
- ↑ U. Müller: Wolframtetrabromidoxid, WOBr4. In: Acta Crystallographica Section C Crystal Structure Communications. 40, pp. 915-917, doi : 10.1107 / S0108270184006259 .

