1,2-dibromooctane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
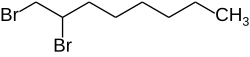
|
||||||||||||||||
| Structural formula without stereochemistry | ||||||||||||||||
| General | ||||||||||||||||
| Surname | 1,2-dibromooctane | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 8 H 16 Br 2 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 272.02 g · mol -1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
1.452 g cm −3 |
|||||||||||||||
| Melting point |
19 ° C |
|||||||||||||||
| boiling point |
241 ° C |
|||||||||||||||
| Vapor pressure |
7.6 Pa (25 ° C) |
|||||||||||||||
| solubility |
1.2 g l −1 in water (25 ° C) |
|||||||||||||||
| Refractive index |
1.493 |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
1,2-Dibromooctane is a doubly brominated aliphatic compound, usually a 1: 1 mixture ( racemate ) of the ( R ) - and the ( S ) - enantiomer . When “1,2-dibromooctane” is mentioned without a prefix in this article or in the scientific literature , the racemate ( RS ) -1,2-dibromooctane is meant.
presentation
Racemic 1,2-dibromooctane can be prepared on a laboratory scale with cooling by electrophilic addition of bromine to 1-octene :
Side reaction
In the event of a homolytic cleavage of the bromine molecule, a radical chain reaction can take place through the bromine radical. This is favored by an increased reaction temperature or high-energy light. Typical by-products are 3-bromo-1-octene from allyl bromination and lower oligomers .
use
1,2-Dibromooctane is used as a synthesis base in the chemical industry. In the EU, the connection was only pre-registered , but not registered .
See also
Individual evidence
- ↑ a b chemnet.com: CAS number: 6269-92-7 1,2-dibromooctane , accessed on November 29, 2015.
- ↑ a b c d Entry on 1,2-dibromooctane in the ChemSpider database of the Royal Society of Chemistry , accessed on November 29, 2015.
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ InfoCard for Octane, 1,2-dibromo from the European Chemicals Agency (ECHA), accessed on January 4, 2019.
