4-hydroxybutyl acrylate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
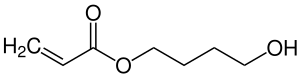
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 4-hydroxybutyl acrylate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 7 H 12 O 3 | |||||||||||||||
| Brief description |
colorless and odorless liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 144.2 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
1.041 g cm −3 |
|||||||||||||||
| Melting point |
−80 ° C |
|||||||||||||||
| boiling point |
236 ° C |
|||||||||||||||
| Vapor pressure |
0.000104 Pa (25 ° C) |
|||||||||||||||
| Refractive index |
1.452 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
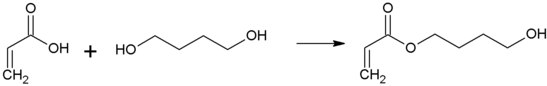
In 4-hydroxybutyl acrylate (4-HBA, 1,4-butanediol monoacrylate) is an ester of acrylic acid . It contains both a hydroxyl group and an unsaturated double bond . It is generally used as a monomer for different classes of binders .
Extraction and presentation
Hydroxybutyl acrylate is produced via the partial esterification of 1,4-butanediol with acrylic acid.
properties
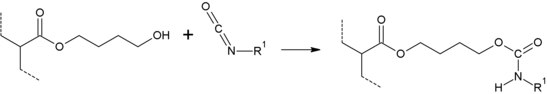
When reacting with isocyanates , hydroxybutyl acrylate has the highest reactivity of the common hydroxyl-containing acrylate monomers. This is based on the one hand on the electron-donating effect of the hydrocarbon chains and on the other hand from the better availability for reaction. This means that a hydroxyl group on a long hydrocarbon chain has a much better mobility and can therefore meet an isocyanate group more easily. It is more reactive than 2-hydroxypropyl acrylate and 2-hydroxyethyl acrylate .
use
Hydroxybutyl acrylate can be incorporated into binders such as polyacrylates via radical or ionic polymerization . Since hydroxybutyl acrylate has a free hydroxyl group, it can be used to incorporate hydroxyl groups into resins. These groups are then available for curing reactions with isocyanates or urea resins .
Individual evidence
- ↑ a b c d e f Entry on 4-hydroxybutyl acrylate in the GESTIS substance database of the IFA , accessed on June 13, 2018(JavaScript required) .
- ↑ a b Data sheet 4-Hydroxybutyl acrylate from Sigma-Aldrich , accessed on June 14, 2018 ( PDF ).
- ↑ a b c Roland Baumstark, Reinhold Schwalm and Manfred Schwartz: Acrylate resins . Vincentz Network, Hannover 2014, ISBN 978-3-86630-820-6 .
- ↑ Ulrich Poth: Synthetic binders for coating systems . Vincentz Network, Hannover 2016, ISBN 978-3-86630-611-0 .