2-hydroxyethyl acrylate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 2-hydroxyethyl acrylate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 5 H 8 O 3 | |||||||||||||||
| Brief description |
colorless liquid with a pungent odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 116.1 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
1.106 g cm −3 |
|||||||||||||||
| Melting point |
<−60 ° C |
|||||||||||||||
| boiling point |
200.3 ° C |
|||||||||||||||
| Vapor pressure |
0.1 h Pa (21.4 ° C) |
|||||||||||||||
| solubility |
miscible with water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
In 2-hydroxyethyl acrylate (HEA) is an ester of acrylic acid . It contains both a hydroxyl group and an unsaturated double bond . It is generally used as a monomer for different classes of binders and polymers.
Extraction and presentation
2-Hydroxyethyl acrylate can be synthesized from the reaction of acrylic acid with ethylene oxide .
If methacrylic acid is used instead of acrylic acid , 2-hydroxyethyl methacrylate is obtained . If hydroxyethyl acrylate is used instead of hydroxyethyl methacrylate in resins and polymers, softer or more flexible properties are obtained.
properties
As a monomer with two different reactive groups, hydroxyethyl acrylate can be used for different applications. If one wishes to synthesize oligomers, resins or polymers with unsaturated double bonds, a reaction can take place via the hydroxyl group. In contrast, oligomers , resins or polymers can be equipped with hydroxyl groups if a polymerization takes place via the unsaturated double bond.
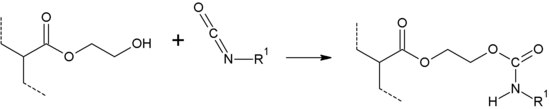
In the reaction with isocyanates , hydroxyethyl acrylate has an average reactivity, which results on the one hand from the electron-donating effect of the hydrocarbon chains and on the other hand from the better availability for the reaction. This means that a hydroxyl group on a long chain has a much better mobility and can thus meet an isocyanate group more easily. It is more reactive than hydroxypropyl acrylate and less reactive than 4-hydroxybutyl acrylate .
use
Hydroxyethyl acrylate can be incorporated into binders such as polyacrylates via free-radical or ionic polymerization . Since hydroxyethyl acrylate has a free hydroxyl group, it can be used to incorporate hydroxyl groups into resins. These groups are then available for curing reactions with isocyanates or urea resins .
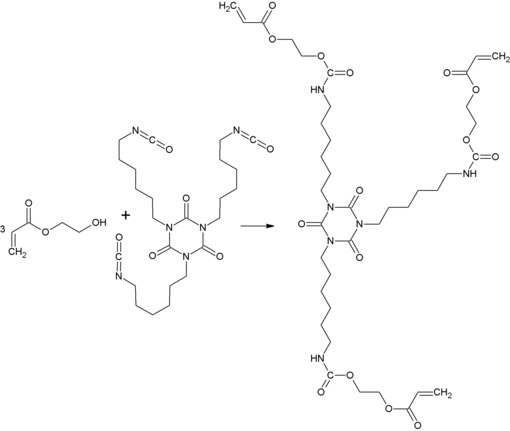
In addition to the possibility of incorporating hydroxyethyl acrylate into resins via the double bond , there is also the possibility of doing this via the hydroxyl group. Example would be this is the reaction of 3 moles of hydroxyethyl acrylate and one mole of HDI - isocyanurate . This results in a urethane acrylate which can be used as a reactive diluent in coatings that can be cured by means of UV radiation .
Individual evidence
- ↑ a b c d e f g h i j Entry on 2-hydroxyethyl acrylate in the GESTIS substance database of the IFA , accessed on June 11, 2018(JavaScript required) .
- ↑ Entry on 2-hydroxyethyl acrylate in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on June 11, 2018. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ a b c d e Baumstark, Roland, Schwalm, Reinhold, Schwartz, Manfred: Acrylate resins . Vincentz Network, Hannover 2014, ISBN 978-3-86630-820-6 .