AL-LAD
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| General | ||||||||||
| Surname | AL-LAD | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 22 H 27 N 3 O | |||||||||
| Brief description |
colorless solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| Drug information | ||||||||||
| Mechanism of action | ||||||||||
| properties | ||||||||||
| Molar mass | 349.47 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
88-89 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
AL-LAD ( English N - al lyl - nor- l ysergic a cid d iethylamide ) is a synthetically produced derivative of lysergic acid , which occurs naturally in ergot alkaloids . Even in very small doses , it produces long-lasting hallucinogenic effects. Pharmacologically, AL-LAD belongs to the group of serotonin-related psychedelic substances.
chemistry
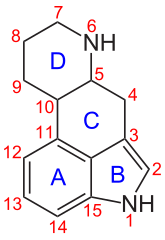
Chemically, AL-LAD belongs to the Ergoline structural class . The tetracyclic ergoline is characteristic of the chemical structure of ergot alkaloids . The ergolines found in nature are methylated on the nitrogen in position 6 of the ergoline system . In contrast to LSD, AL-LAD has an N 6 - allyl group instead of an N 6 - methyl group .
Effect on humans

The duration of an uncomplicated AL-LAD experience is usually between six and eight hours, depending on the dosage, body weight and age. The effective dose is given as 80–160 µg . AL-LAD acts on the receptor subtypes of the serotonin (5-HT 2 ) receptors and the dopamine receptors .
See also
literature
- Alexander Shulgin , Ann Shulgin: TiHKAL, the Continuation . Transform Press, Berkeley 1997, ISBN 0-9630096-9-9 . ( online )
- VJ Watts, RB Mailman, CP Lawler, KA Neve, DE Nichols: LSD and structural analogs: pharmacological evaluation at D1 dopamine receptors. In: Psychopharmacology. 118, 1995, p. 401, doi : 10.1007 / BF02245940 .
- Andrew J. Hoffman, David E. Nichols: Synthesis and LSD-like discriminative stimulus properties in a series of N (6) -alkyl norlysergic acid N, N-diethylamide derivatives. In: Journal of Medicinal Chemistry. 28, 1985, p. 1252, doi : 10.1021 / jm00147a022 .
- T. Niwaguchi, Y. Nakahara, H. Ishii: Studies on lysergic acid diethylamide and related compounds. IV. Syntheses of various amide derivatives of norlysergic acid and related compounds. In: Yakugaku Zasshi. Volume 96, Number 5, May 1976, pp. 673-678, ISSN 0031-6903 . PMID 987200 .
- Robert C. Pfaff, Xuemei Huang, Danuta Marona-Lewicka, Robert Oberlender and David E. Nichols: Lysergamides Revisited. In: NIDA Research Monograph 146: Hallucinogens: An Update. P. 52, 1994, United States Department of Health and Human Services .
- SD Brandt, PV Kavanagh, F. Westphal, SP Elliott, J. Wallach, T. Colestock, TE Burrow, SJ Chapman, A. Stratford, DE Nichols, AL Halberstadt: Return of the lysergamides. Part II: Analytical and behavioral characterization of N (6) -allyl-6-norlysergic acid diethylamide (AL-LAD) and (2'S, 4'S) -lysergic acid 2,4-dimethylazetidide (LSZ). In: Drug testing and analysis. June 2016, doi : 10.1002 / dta.1985 , PMID 27265891 . Open access: 1H and 13C NMR spectra of N6-allyl-6-norlysergic acid diethylamide (AL-LAD). In: BLOTTER, 15 Jul 2016, 2 (1). doi : 10.16889 / isomerdesign-3
Web links
Individual evidence
- ↑ a b c Alexander and Ann Shulgin: AL-LAD . In: TiHKAL. Transform Press, Berkeley 1997, ISBN 0-9630096-9-9 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ VJ Watts, RB Mailman, CP Lawler, KA Neve, DE Nichols: LSD and structural analogs: pharmacological evaluation at D1 dopamine receptors. In: Psychopharmacology. 118, 1995, pp. 401-409, doi : 10.1007 / BF02245940 .