Dichlorotoluenes
Dichlorotoluenes (dichloromethylbenzenes) form a group of substances that are derived from both toluene and chlorobenzene . The structure consists of a benzene ring with an attached methyl group (–CH 3 ) and two chlorine atoms (–Cl) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 7 H 6 Cl 2 . Dichlorotoluenes therefore belong to the polychlorinated toluenes.
properties
The dichlorotoluenes are practically insoluble in water; however, they are in other organic solvents, such as. B. benzene, alcohol, ether, ethyl acetate and chloroform, soluble.
| Dichlorotoluenes | |||||||||||||
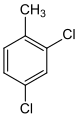
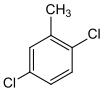
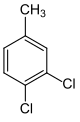
| Surname | 2,3-dichlorotoluene | 2,4-dichlorotoluene | 2,5 dichlorotoluene | 2,6 dichlorotoluene | 3,4-dichlorotoluene | 3,5 dichlorotoluene | |||||||
| other names | 1,2-dichloro- 3-methylbenzene |
1,3-dichloro- 4-methylbenzene |
1,4-dichloro- 3-methylbenzene |
1,3-dichloro- 2-methylbenzene |
1,2-dichloro- 4-methylbenzene |
1,3-dichloro- 5-methylbenzene |
|||||||
| Structural formula |

|
|
|

|
|

|
|||||||
| CAS number | 32768-54-0 | 95-73-8 | 19398-61-9 | 118-69-4 | 95-75-0 | 25186-47-4 | |||||||
| 29797-40-8 (mixture of isomers) | |||||||||||||
| ECHA InfoCard | 100.046.534 | 100.002.224 | 100.039.105 | 100,003,882 | 100.002.226 | 100,042,460 | |||||||
| 100.045.307 (mixture of isomers) | |||||||||||||
| PubChem | 34702 | 7254 | 29572 | 8368 | 7256 | 32834 | |||||||
| Molecular formula | C 7 H 6 Cl 2 | ||||||||||||
| Molar mass | 160.95 g mol −1 | ||||||||||||
| Physical state | liquid | ||||||||||||
| Brief description | colorless liquid | colorless, flammable liquid with an aromatic odor |
colorless liquid | colorless, flammable liquid with an aromatic odor |
colorless liquid with a characteristic odor |
||||||||
| Melting point | 6 ° C | −13 ° C | 4-5 ° C | 2 ° C | −15 ° C | ||||||||
| boiling point | 207-208 ° C | 201 ° C | 197-200 ° C | 196-203 ° C | 200-202 ° C | ||||||||
| density | 1.23 g / cm 3 | 1.25 g / cm 3 | 1.25 g / cm 3 | 1.27 g / cm 3 | 1.25 g / cm 3 | ||||||||
| solubility | practically insoluble in water | ||||||||||||
|
GHS labeling |
|
|
|
|
|
|
|||||||
| H and P phrases | 332 | 411 | 332 | 315-411 | 411 | see above | |||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | see above | ||||||||
| no P-phrases | 273 | no P-phrases | no P-phrases | 262-273 | see above | ||||||||
| Tox data | 2400 mg kg −1 ( LD 50 , rat , oral ) | ||||||||||||
Extraction and presentation
The dichlorotoluenes can be obtained by chlorinating chlorotoluenes , which can be prepared from the toluidines by means of the Sandmeyer reaction .
use
Dichlorotoluenes are used, among other things, as a cleaning agent in the shipping industry, as a dye carrier in the textile industry and as a barrier liquid in vacuum pumps. 2,4-dichlorotoluene is a precursor for the production of 2,4-dichlorobenzoyl chloride and 2,4-dichlorobenzoic acid , which in turn is used for the production of dyes, pharmaceuticals and preservatives. 3,4-dichlorotoluene is used as an intermediate in the production of 3,4-dichlorobenzyl chloride (a precursor for disinfectants), 3,4-dichlorobenzaldehyde (a precursor for dyes) and 3,4-dichlorobenzoic acid .
See also
- Chlorotoluenes
- Chlorobenzyl chlorides
- Trichlorotoluenes , C 7 H 5 Cl 3
- Tetrachlorotoluenes , C 7 H 4 Cl 4
- Pentachlorotoluene , C 7 H 3 Cl 5
safety instructions
Chlorotoluenes can decompose plastics and slightly corrode metals. The thermal decomposition of chlorotoluenes forms toxic and corrosive hydrogen chloride .
Individual evidence
- ↑ Entry for CAS no. 29797-40-8 in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d e f data sheet 2,3-dichlorotoluene from Sigma-Aldrich , accessed on December 27, 2019 ( PDF ).
- ↑ a b c d e f g Entry on 2,4-dichlorotoluene in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d e f data sheet 2,5-dichlorotoluene from Sigma-Aldrich , accessed on December 27, 2019 ( PDF ).
- ↑ a b c d e f Entry on 2,6-dichlorotoluene in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d e f Entry on 3,4-dichlorotoluene in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ^ Association of authors: Organikum , 19th edition, Johann Ambrosius Barth, Leipzig · Berlin · Heidelberg 1993, ISBN 3-335-00343-8 , p. 566.
- ↑ Toxicological assessment of dichlorotoluene, technical mixture of isomers (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.
- ↑ Toxicological assessment of 2,4-dichlorotoluene (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.
- ↑ Toxicological assessment of 3,4-dichlorotoluene (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.

