Diethylthiamobutene
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||
| Simplified structural formula without stereochemistry | |||||||||||||
| General | |||||||||||||
| Non-proprietary name | Diethylthiamobutene | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 16 H 21 NS 2 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| Drug information | |||||||||||||
| Drug class | |||||||||||||
| properties | |||||||||||||
| Molar mass | 291.47 g · mol -1 (Diethylthiambuten) | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Diethylthiambutene , or simply thiambutene , is a fully synthetic opioid with a strong analgesic effect. Diethylthiambutene is a pure agonist at the μ-opioid receptor . It is the basic structure of a number of opioids ( thiambutenes ), which include , for example, dimethylthiambutene , ethylmethylthiambutene , pyrrolidinylthiambutene and piperidylthiambutene .
Overview reaction
Extraction and presentation
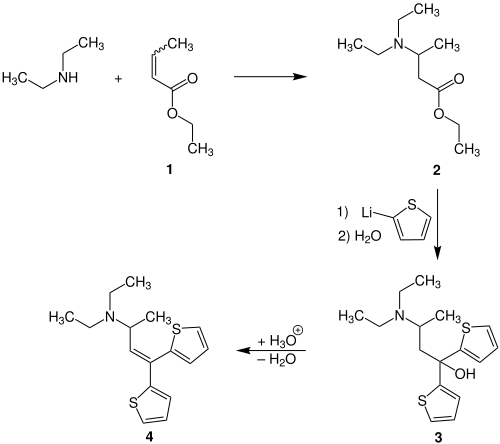
Starting from diethylamine and Crotonsäureethylester 1 and subsequent Grignard reaction is the intermediate product 2 to the alcohol 3 synthesized . Acid-catalyzed dehydration then gives diethylthiamobutene 4 .
history
Diethylthiambutene was developed as a potent analgesic by Burroughs Wellcome & Company in 1950 . The hydrochloride of diethylthiambutene is used medicinally. Diethylthiambutene cannot be prescribed in Germany.
pharmacology
effect
As an opioid, diethylthiambutene has the same profile of effects and side effects, and thus essentially the same risk potential as other opioids.
Side effects
Side effects can occur with diethylthiambutene that exceeds the pre-existing tolerance to opioids due to the pre-consumption. These are tiredness, sleep disturbances , drowsiness , nausea , vomiting , edema in the legs, urinary retention , constipation and pruritus . They usually go away as tolerance develops or the dose is reduced. Sleep and sexual disorders last the longest .
- Use during pregnancy and breastfeeding
Diethylthiambutene has an effect on the fetus when taken during pregnancy.
Trade names
Monopreparations : Themalon.
literature
- The Merck Index : An Encyclopedia of Chemicals, Drugs, and Biologicals . 15th edition. Merck & Co., Whitehouse Station NJ 2013, ISBN 978-1-849736-70-1 , p. 1722.
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ DW Adamson: 180. Aminoalkyl tertiary carbinols and derived products. Part II. 3-Amino-1: 1-di-2'-thienyl-alkan-1-ols and -alk-1-enes . In: J. Chem. Soc. January 1950, p. 885-890 , doi : 10.1039 / JR9500000885 .
- ↑ Patent US2561899 : Dithienyl allyl amines. Published July 24, 1951 , Applicant: Burroughs Wellcome Co., Inventor: Wallace Adamson Donald.