Disodium methyl arsonate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
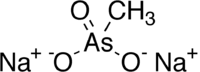
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Disodium methyl arsonate | |||||||||||||||
| other names |
DSMA |
|||||||||||||||
| Molecular formula | CH 3 AsNa 2 O 3 | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 183.93 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
132-139 ° C |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Disodium methyl arsonate is a chemical compound of sodium from the group of arsonates or the organic arsenic compounds . It is the disodium salt of methylarsonic acid .
Extraction and presentation
Disodium methyl arsonate can be obtained by reacting sodium arsenite with methyl iodide in the presence of alkali hydroxides such as sodium hydroxide or by reacting sodium arsenite with dimethyl sulfate .
properties
Disodium methyl arsonate is a colorless solid that is easily soluble in water.
use
Disodium methyl arsonate (like sodium methyl arsenate ) is used as a herbicide (in the USA for cotton). It has also been used in veterinary medicine.
Individual evidence
- ^ A b Christopher G. Morris, Academic Press: Academic Press Dictionary of Science and Technology . Gulf Professional Publishing, 1992, ISBN 0-12-200400-0 , pp. 659 ( limited preview in Google Book search).
- ↑ a b c d e f g Entry on disodium methyl arsonate in the GESTIS substance database of the IFA , accessed on March 7, 2020(JavaScript required) .
- ↑ Terry Hills: Lotus Illustrated Dictionary of Organic Chemistry . Lotus Press, 2004, ISBN 978-81-89093-51-8 , pp. 68 ( limited preview in Google Book search).
- ↑ a b c P. H. List, L. Hörhammer: Chemicals and Drugs Part A: NQ . Springer-Verlag, 2013, ISBN 978-3-642-65035-2 , pp. 108 ( limited preview in Google Book search).
- ↑ Land Use Effect on Arsenic Concentrations in Benton and Washington Counties . ProQuest, 2008, ISBN 0-549-66748-2 , pp. 12 ( limited preview in Google Book search).
- ↑ Andrew D. Eaton, Hsaio Chiu Wang, Jack Northington, AWWA Research Foundation: Analytical Chemistry of Arsenic in Drinking Water . American Water Works Association, 1998, ISBN 978-0-89867-907-6 , pp. 2 ( limited preview in Google Book search).



