GV (nerve agent)
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||
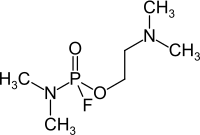
| Simplified structural formula without stereochemistry | ||||||||||
| General | ||||||||||
| Surname | GV | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 6 H 16 FN 2 O 2 P | |||||||||
| Brief description |
colorless, highly viscous liquid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 198.18 g mol −1 | |||||||||
| Physical state |
liquid |
|||||||||
| density |
1.11 g cm −3 (25 ° C) |
|||||||||
| Melting point |
−110 ° C |
|||||||||
| boiling point |
226 ° C |
|||||||||
| Vapor pressure |
6.5 Pa (25 ° C) |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| Toxicological data |
11–1222 µg kg −1 ( LD 50 , rodent , transdermal ) |
|||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
GV is the name of a Soviet nerve agent , which is structurally similar to the neurotoxin soman . The US Department of Defense and NATO gave this warfare agent the designation GV (G for Germany ; V for English viscous "tough, viscous").
Isomerism
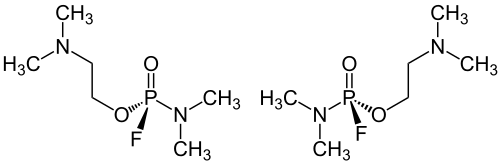
GV has a stereocenter on the phosphorus atom, so there are two enantiomers , one has an ( R ) configuration, the other has an ( S ) configuration. The manufacturing processes described provide the racemic GV, i.e. a 1: 1 mixture of ( R ) - (dimethylamido) fluorophosphoric acid 2- N ', N ' - (dimethylaminoethyl) ester (left) and ( S ) - (dimethylamido) fluorophosphoric acid- 2- N ′, N ′ - (dimethylaminoethyl) ester (right):
Protective measures
Even the smallest amounts of GM are fatal. The target is the entire body. Therefore, only a full-body protective suit and a protective mask offer sufficient protection. Oxime tablets can be taken before a warfare agent is used. During the weeks of post-treatment, trying to Obidoxime the acetylcholinesterase regenerate.
Oxidizing agents and alkaline solutions can be used for decontamination , as the hydrolysis of nerve agents is accelerated in an alkaline environment. Chlorinated lime for insensitive surfaces and sodium carbonate solutions for sensitive objects have also proven their worth .
See also
Individual evidence
- ↑ a b c d e f Steven L. Hoenig: Compendium of chemical warfare agents . Springer, New York NY 2007, ISBN 978-0-387-34626-7 ( limited preview in Google Book Search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.