Potassium propionate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
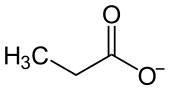 |
||||||||||||||||
| General | ||||||||||||||||
| Surname | Potassium propionate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 3 H 5 KO 2 | |||||||||||||||
| Brief description |
colorless, crystalline platelets |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 112.17 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
> 300 ° C |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Potassium propionate is the potassium salt of propionic acid . The chemical compound is used in the food industry as a preservative under the number E 283 , especially in sliced bread and baked goods . The hygroscopic salt crystallizes from aqueous solutions as a monohydrate (C 2 H 5 COOK · H 2 O).
properties
Potassium propionate crystallizes as a monohydrate, which gives off its water of crystallization at 120 ° C.
safety instructions
With prolonged administration of propionic acid and propionates in the feed of rats in doses between 0.6 and 5%, these cause changes in the forestomach ( thickening and inflammation). However, this is classified as a species-specific reaction for rats, since no such effects were observed in other animal species such as mice and rabbits.
Situation in Germany
The use of propionic acid or its sodium , potassium or calcium salt in food was banned in the Federal Republic of Germany from January 1, 1988, as no longer a need for use was seen. In the EU, however, its use is permitted again.
Individual evidence
- ↑ a b c d J. IG Cadogan, J. Buckingham, F. MacDonald: Dictionary of Organic Compounds: First Supplement. CRC Press, 1996, ISBN 978-0-412-54110-0 .
- ↑ a b c A. Renard: "Sur les propionates métalliques" in Comptes rend. hebd. 1887 , 104 , pp. 913-917 ( full text ).
- ↑ There is not yet a harmonized classification for this substance . A labeling of potassium propionate in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), retrieved on July 28, 2019, is reproduced from a self-classification by the distributor .
- ^ W. Baltes: Food chemistry . 3rd ed., Springer, Berlin et al., 1992. pp. 160-161.
- ↑ a b H.-G. Classen, PS Elias, WP Hammes, M. Winter: Toxicological-hygienic assessment of food ingredients and additives. Behr's Verlag, 2001, ISBN 978-3-86022-806-7