Wedge formula
The wedge line formula (English Natta Projection after Giulio Natta ) is one of the chemical structural formulas . It clarifies the spatial arrangement of the molecular components (the substituents ). This communication and knowledge of the spatial arrangement is of considerable practical importance, especially in the case of chiral medicinal substances , since the stereoisomers usually develop different pharmacological effects in the body.
| Structural formulas | Other modes of representation | ||||||
|---|---|---|---|---|---|---|---|
| Electron formula | Valence stroke formula | Wedge formula | Skeletal formula | Constitutional formula | Molecular formula | Ratio formula | |
| methane | does not exist | CH 4 | CH 4 | CH 4 | |||
| propane |

|

|

|
|
CH 3 -CH 2 -CH 3 | C 3 H 8 | C 3 H 8 |
| acetic acid |

|

|

|

|
CH 3 -COOH | C 2 H 4 O 2 | CH 2 O |
| water | does not exist | does not exist | H 2 O | H 2 O | |||
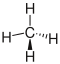
The use of the wedge line formula, also known as wedge line notation, is particularly helpful to clarify the stereochemistry of a molecule, as the absolute configuration can also be recognized in the two-dimensional plane of the drawing. A filled wedge indicates that the remainder (or the substituent or the atom) protrudes from the plane of the drawing; a dashed wedge or dashed line indicates that the substituent is behind the plane of the drawing. Bindings that lie in the drawing plane are drawn as simple lines. In methane (bond angle H – C – H: 109 ° 28 '), for example
- two carbon-hydrogen bonds in the plane of the drawing (single line),
- a carbon-hydrogen bond protrudes from the plane of the drawing (wedge-shaped, bold bond) and
- a carbon-hydrogen bond is located behind the plane of the drawing (bond shown in dashed lines).
The wedge line presentation is rarely used separately, but mostly combined with a skeletal formula .
See also
Individual evidence
- ↑ Everhardus Ariëns : Stereochemistry, a basis for sophisticated nonsense in pharmacokinetics and clinical pharmacology , European Journal of Clinical Pharmacology 26 (1984) 663-668, doi : 10.1007 / BF00541922 .
- ↑ Hermann J. Roth, Christa E. Müller, Gerd Folkers: Stereochemie und Arzneimittel, Wissenschaftliche Verlagsgesellschaft Stuttgart, 1998, pp. 33-104, ISBN 3-8047-1485-4 .
- ↑ K. Peter C. Vollhardt: Organische Chemie , VCH, 1990, pp. 31-32, ISBN 3-527-26912-6 .
- ↑ Harold Hart: Organische Chemie , VCH, 1983, p. 30, ISBN 3-527-26480-9 .
- ↑ Ivan Ernest: Binding, Structure and Reaction Mechanisms in Organic Chemistry , Springer-Verlag, 1972, p. 5, ISBN 3-211-81060-9 .