Methoxybenzaldehydes
| Methoxybenzaldehydes | ||||||
| Surname | 2-methoxybenzaldehyde | 3-methoxybenzaldehyde | 4-methoxybenzaldehyde | |||
| other names |
o -methoxybenzaldehyde |
m -methoxybenzaldehyde |
p -methoxybenzaldehyde anisaldehyde |
|||
| Structural formula |  |
 |
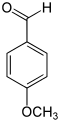
|
|||
| CAS number | 135-02-4 | 591-31-1 | 123-11-5 | |||
| PubChem | 8658 | 11569 | 31244 | |||
| Molecular formula | C 8 H 8 O 2 | |||||
| Molar mass | 136.15 g mol −1 | |||||
| Physical state | firmly | liquid | ||||
| Brief description | yellowish liquid with a pleasant odor |
yellowish liquid with a characteristic odor |
||||
| density | 1.127 g cm −3 | 1.12 g cm −3 | 1.12 g cm −3 | |||
| Melting point | 34-40 ° C | 3-4 ° C | 0-2 ° C | |||
| boiling point | 238 ° C | 230-233 ° C | 247-249 ° C | |||
| solubility | Practically insoluble in water, soluble in ethanol and ether | |||||
|
GHS labeling |
|
|
|
|||
| H and P phrases | no H-phrases | no H-phrases | no H-phrases | |||
| no P-phrases | no P-phrases | no P-phrases | ||||
In chemistry, methoxybenzaldehydes form a group of substances that are derived from both benzaldehyde and anisole . The structure consists of a benzene ring with attached aldehyde (-CHO) and methoxy groups (-OCH 3 ) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 8 H 8 O 2 . First and foremost, they can be viewed as methoxy-substituted benzaldehydes. 4-Methoxybenzaldehyde is mainly known by its common name anisaldehyde . In the English-language literature, the term anisaldehyde ( anisaldehyde ) is also used for the 2- and 3-methoxybenzaldehydes.
Individual evidence
- ↑ a b c d e Entry on 3-methoxybenzaldehyde in the GESTIS substance database of the IFA , accessed on April 3, 2019(JavaScript required) .
- ↑ a b c d e f Entry on 4-methoxybenzaldehyde in the GESTIS substance database of the IFA , accessed on April 3, 2019(JavaScript required) .
- ↑ a b c d data sheet 2-Methoxybenzaldehyde from Sigma-Aldrich , accessed on April 3, 2019 ( PDF ).
- ↑ CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
- ↑ Entry on o-anisaldehyde at ChemBlink , accessed on December 28, 2019.
Web links
Commons : Methoxybenzaldehyde - Collection of images, videos and audio files
- Freepatentsonline: Process for preparing a methoxybenzaldehyde from the corresponding phenolic benzaldehyde
See also
- Methoxybenzyl alcohols
- Methoxybenzoic acids
- Methoxyacetophenone C 9 H 10 O 2 (3 isomers e.g. 4-methoxyacetophenone )
Derivatives
- Hydroxymethoxybenzaldehyde C 8 H 8 O 3 (10 isomers, e.g. vanillin = 4-hydroxy-3-methoxybenzaldehyde, isovanillin = 3-hydroxy-4-methoxybenzaldehyde and ortho-vanillin = 2-hydroxy-3-methoxybenzaldehyde)
- Fluoromethoxybenzaldehydes C 8 H 7 FO 2 (10 isomers, with di- and trifluorides as well as the corresponding chloro- and bromomethoxybenzaldehydes exist)